Minkowski-Choffard disease is a common hereditary disease belonging to the group of anemias. Its other names are hereditary spherocytosis and microspherocytic hemolytic anemia. The pathology was first described at the end of the 19th century by the Polish physiologist Minkowski, then supplemented by the French doctor Chauffard.
People of all ethnic groups are susceptible to the disease, but it is more often diagnosed in European territory, especially its northern part. It can appear for the first time at any age.
Anemia - what is it?
Anemia is characterized by a decrease in the level of red blood cells and hemoglobin in the blood. In some cases, not only the numerical composition of red blood cells falls below normal, but their shape also changes. As the disease progresses, the red blood cells are unable to perform their functions.
Anemia develops against the background of other disorders in the body; it does not manifest itself. Therefore, to get rid of the disease, it is necessary to establish the cause that led to the change in the composition of the blood.
Classification of the disease
There are 5 main types of anemia:
Posthemorrhagic
Develops as a result of severe blood loss caused by injuries, surgical interventions, ectopic pregnancy, peptic ulcer, diseases of the blood, lungs, and liver. Leads to vascular insufficiency and oxygen starvation.
For an adult, a serious health threat occurs when a person loses 500 milliliters of blood.
Scarce
Caused by a deficiency of vitamins or minerals necessary for hematopoiesis.
There are 3 most common types of deficiency anemia:
Iron deficiency
The most common type. Diagnosed in 80% of cases. Occurs when there is a small amount of iron elements in the body.
B12 deficiency (megaloblastic, pernicious, Addison-Beermer disease)
It develops when the body does not have enough vitamin B12 (cyanocobalamin). A similar situation occurs when there is a failure in the synthesis of the glycoprotein that controls the amount of vitamin B12.
The disease is often diagnosed in older people, patients with atrophy of the gastric mucosa, enteritis, and rarely in pregnant women.
Folate deficiency
Characterized by a deficiency of vitamin B9 (folic acid)
Hemolytic
A lack of red blood cells is observed due to their intensive destruction. This may increase the concentration of free hemoglobin, hemosiderin, and bilirubin. To compensate for the losses, the body begins to actively produce new red blood cells, but they will be immature and cannot fully perform their assigned function.
Hemolytic anemia can be congenital (caused by gene defects) or acquired. It can develop due to incompatibility of the blood of a pregnant woman and a child, intoxication of the body, transfusion of inappropriate blood, taking a number of medications, or the presence of malignant neoplasms.
The most common ancestral varieties are:
Sickle cell anemia
A characteristic feature is the presence in the blood of defective red blood cells, shaped like a crescent. Not only can they not do their job, but they can also clog blood vessels.
Thalassemia
It is characterized by the target-like shape of red blood cells.
Minkowski-Shoffar
It is characterized by spherocytosis of red blood cells
Aplastic
The result of suppression of the hematopoietic capacity of the bone marrow as a result of the use of drugs, immune pathologies, the influence of chemicals, ionizing radiation, etc. In addition to a decrease in the level of red blood cells, there is a decrease in the number of platelets, leukocytes and bone marrow cells. Therefore, aplastic anemia is often accompanied by thrombocytopenia, anemia, and leukopenia.
Aplastic anemia can be hereditary or acquired.
Sideroachrestic
The disease is hereditary and mainly occurs in men. The hemoglobin content in red blood cells decreases, and free iron accumulates in the body
Anemia is also distinguished:
- microcytic - red blood cells are small, contain little hemoglobin;
- normocytic - red blood cells are of normal size, but their number is reduced;
- macrocytic - red blood cells are large, but they contain little hemoglobin.
What is the distinctive feature of hemolytic anemia?
Hemolytic anemia combines several diseases, but the reasons for their development are the same. Red blood cells in the blood are destroyed too quickly, so the red bone marrow begins to produce them intensively. As a result, their growth cycle is disrupted. As anemia progresses, red blood cells undergo massive death.
Hemolytic anemia can be transmitted from parents to children, and can develop throughout life. Minkowski-Choffard anemia is a hereditary pathology.
This disease is also called microspherocytic anemia and hereditary spherocytosis. However, the term “Minkowski-Choffard anemia” is most often used after the scientists who first discovered this pathology.
This type of anemia cannot be called rare; it affects every 5,000th person on the planet. Most often it affects people living in Northern Europe. Minkowski-Choffar anemia first makes itself felt in childhood. If this disorder is left untreated, the body as a whole will suffer.
Causes of Minkowski-Choffard anemia
With Minkowski-Choffard anemia, the structure of red blood cells undergoes pathological changes, which affects their functioning. Red blood cells become fragile and are destroyed much more easily. Hemoglobin comes out of them, which circulates in free form in the blood.
If normally red blood cells have the shape of a biconvex disk, then with Minkowski-Choffard anemia they become round, which does not allow them to penetrate small blood vessels. When attempting to do this, the red blood cells are severely damaged and destroyed. Their level in the blood decreases, which causes the development of anemia.
If one of the parents suffered from Minkowski-Choffard anemia, then this disease will be inherited by the child. Rarely sick children are born from completely healthy parents. If this happens, experts say the cause is damage to the DNA structure. Genes mutate while the baby is in the womb.
Pathogenic factors are:
- Exposure of a woman’s body to radioactive or x-ray radiation.
- Poisoning of a pregnant woman’s body with salts of heavy metals, drugs, and tobacco smoke.
- Transmitted viral diseases.
It should be taken into account that these pathogenic factors can provoke not only anemia, but also other serious disorders in the body of a growing fetus. Therefore, you need to treat your health very carefully.
Symptoms of Minkowski-Choffard anemia
The lower the level of normal red blood cells in the blood, the stronger the symptoms of the disease will be. The first signs of pathology develop in a child in the preschool period or during early school age. Anemia has a wave-like course, periods of remission are followed by periods of exacerbation.
During a hemolytic crisis, massive death of red blood cells occurs. At the same time, all the symptoms gain strength. When the crisis is over, the person will feel satisfactory. Possible excessive pallor of the skin and mucous membranes.
Symptoms of a crisis are:
- High body temperature.
- Headache.
- General weakness and malaise.
- Yellowness of the skin.
- Cramps and pain in the abdomen.
- Enlargement of the liver.
- Inflammation of the spleen.
The disease can also develop in adulthood. The first sign that a person most often pays attention to is yellowing of the skin. Although in some cases, objective symptoms of the disorder may be completely absent. Often, anemia can be diagnosed only during a random examination, when a person donates blood for analysis.
The article provides a review of the literature concerning clinical and laboratory manifestations, diagnosis, treatment and prognosis of hereditary microspherocytic anemia. The results of our own observations of two patients undergoing treatment in the oncohematology department of the Yoshkar-Ola Children's City Hospital are described.
Hereditary microspherocytosis (Minkowsky - Shauffard disease)
The paper presents a literature review of clinical and laboratory manifestations, diagnosis, treatment and prognosis of hereditary microspherocytic anemia. It is describes the results of his own observations of two patients treated in oncohematological office in Yoshkar-Ola City Children's Hospital.
Hereditary microspherocytosis was first described in 1900 by Minkowski, and later in more detail by Shoffar. The prevalence of this disease is 1:5000 of the population, most often found in residents of Northern Europe [1, 2].
Pathogenesis. Congenital microspherocytic hemolytic anemia is a familial disease inherited in an autosomal dominant manner. The disease is based on a genetic defect in the protein of the erythrocyte membrane, as a result of which its permeability to sodium ions increases, which leads to swelling of erythrocytes, disruption of the ability of erythrocytes to deform, detachment of part of their surface in the spleen, shortening of their life span and destruction by spleen macrophages [3, 4 ]. The pathology of erythrocytes is manifested by a morphological abnormality - microspherocytosis. The duration of stay of microspherocytes in the circulating blood is sharply reduced; the average period of their stay in the bloodstream can be 12-14 days (instead of the normal 120-125 days) [5].
Clinical picture. The central place in the clinical picture belongs to the hemolytic syndrome, which is manifested by three cardinal signs: jaundice, splenomegaly and anemia [5]. Signs of delayed development may be observed, as well as disturbances of the facial skeleton in the form of a “tower skull”, saddle nose, high palate, disturbance of the arrangement of teeth, and narrow eye sockets [6]. The severity of anemic syndrome varies. A moderate decrease in hemoglobin is often observed. Some patients have no anemia at all. The most dramatic anemization is observed during the hemolytic crisis. Microspherocytic hemolytic anemia has a chronic course and is accompanied by periodic crises and remissions. Hemolytic crisis occurs under the influence of provoking factors (infection, hypothermia, overwork, pregnancy and others) and is manifested by a sharp increase in symptoms against the background of continuously ongoing hemolysis [6]. At the same time, the temperature rises due to the massive breakdown of red blood cells, the intensity of jaundice increases, hepatomegaly is pronounced, the spleen is dense and smooth, often painful as a result of tension of the capsule during blood filling or perisplenitis. Hemolytic disease is often complicated by attacks of hepatic colic, due to the formation of pigment stones in the gall bladder and bile ducts. Due to attacks of hepatic colic and stagnation of bile in the liver, patients may experience symptoms of angiocholecystitis and parenchymal hepatitis with the appearance of direct bilirubin in the blood. With exacerbation of the disease, there is a tendency to nosebleeds [3, 7]. Despite its congenital nature, the disease only rarely manifests itself in the first days after birth; symptoms usually appear in childhood, usually at 3-10 years of age, or adulthood [8].
Laboratory diagnostics. Microscopic examination of peripheral blood preparations reveals spherocytes, characterized by a decrease in the average diameter (less than 7.2-7.0 µm) against the background of a normal average MCV volume and an increased MCHC value. The red blood cell size distribution curve (Price-Jones curve) on the graph is shifted to the left. Anemia is normochromic in nature. The RDW value exceeds 12% (anisocytosis). Reticulocytosis - 15%-30% [1, 10]. The white blood cell count is usually normal. In hemolytic crises, neutrophilic leukocytosis with a shift to the left is noted. The platelet count varies within normal limits. The content of indirect bilirubin in the blood is moderately increased and, as a rule, does not exceed 50-70 µmol/l [6, 7]. Autoantibodies are not detected on the erythrocyte membrane; the direct Coombs test is negative. A characteristic laboratory sign is a decrease in the osmotic resistance of red blood cells to hypotonic sodium chloride solutions. The beginning of hemolysis in hereditary microspherocytosis corresponds to 0.6-0.7%, and the end - 0.4% instead of 0.48-0.22% normally [1, 9]. A decrease in osmotic resistance indicates the predominance of spherical erythrocytes in the blood - spherocytes, which are less resistant to osmotic hemolysis than normal macroplanocytes (Fig. 1).
Figure 1. Morphology of microspherocytes in peripheral blood smears of a patient (left), healthy (right)
Differential diagnosis of hereditary microspherocytosis comes down to the diagnosis of hemolytic anemia in general. Many patients are mistakenly diagnosed with Gilbert's syndrome, chronic hepatitis or even cirrhosis of the liver, and anemia is considered a consequence of these diseases. In all cases of jaundice, a thorough examination of the patient is necessary, regardless of hemoglobin content. A predominant increase in the content of indirect bilirubin, reticulocytes, and microspherocytosis detected when viewing a smear provide the basis for the correct diagnosis. A differential diagnosis of autoimmune hemolytic anemia and microspherocytosis is then necessary, since autoimmune hemolytic anemias often produce symptomatic microspherocytosis [3]. In this situation, a thorough ascertainment of the medical history, duration of the disease, the presence of a similar disease in relatives, identification of skeletal changes, and testing to detect autoantibodies (direct Coombs test) help.
Treatment and prognosis. Treatment during a hemolytic crisis is aimed at eliminating anemia, hypoxia, and hyperbilirubinemia. The method of choice for the treatment of hereditary spherocytosis is splenectomy, optimal at the age of 4-5 years. Splenectomy provides practical recovery, despite the preservation of spherocytosis. Indications for splenectomy in microspherocytosis are persistent anemia or that occurs in the form of crises, significant hyperbilirubinemia, even without anemia, the appearance of pain in the right hypochondrium, and developmental delays in children [3]. Blood (packed red blood cell) transfusions are indicated in cases of severe hemolytic crisis [5]. The prognosis for hereditary microspherocytosis is favorable. The probability of developing the disease in children if one of the spouses has microspherocytosis is slightly lower than 50% [9].
Clinical case No. 1. Patient S., 1 year 10 months, from 06/01/09 to 06/09/09 was undergoing an inpatient examination in the oncohematology department of the Yoshkar-Ola Children's City Hospital. From the anamnesis it is known that the child from the first pregnancy was born premature, weighing 900 grams. He was nursed in the department of the second stage of nursing premature babies. Heredity is unknown, since he was adopted at the age of four months. From birth, a decrease in the level of hemoglobin in the blood test and icterus of the skin were noted. Since December 2008, he has been under observation by a hematologist for anemia of prematurity and has been taking iron supplements. Upon receipt of a complaint of lethargy, weakness, fever to febrile levels, pallor and icteric discoloration of the skin. On palpation, the liver protrudes from under the edge of the costal arch by 2.5 cm, the spleen by 2 cm, is soft and elastic.
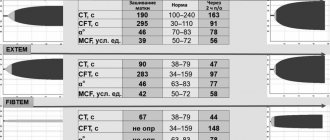
General blood test dated 06/01/09: HGB - 106 g/l, RBC - 3.9×10¹²/l, WBC - 11.6×109/l, Ht - 26%, MCV - 68.8 fl, MCH — 25.8 pg, MSHC — 375 g/l, RDW — 20.5%, PLT — 434×109/l, ESR — 6 mm/h, in the leukocyte formula: eosinophils — 10%, neutrophils — 34 %, lymphocytes - 47%, monocytes - 9%.
Anemia is normochromic in nature. In blood smears, microspherocytes predominate, characterized by the absence of central clearing characteristic of normal erythrocytes, which is confirmed graphically on the Price-Jones curve, reflecting the quantitative ratios of erythrocytes of different diameters. The apex of the Price-Jones curve is stretched and shifted to the left towards microcytes. In addition, normocytes and single macrocytes are visible in blood smears, this confirms an increase in the RDW index - 20.5%. In many red blood cells, basophilic punctation and polychromatophilia are detected. Reticulocytosis - 27%. Osmotic resistance of erythrocytes: min - 0.52%, max - 0.34% (with a norm of 0.48-0.22%) - a decrease in resistance to hypotonic solutions of table salt is determined. Biochemical analysis dated 06/02/09: concentration of indirect bilirubin - 29.9 µmol/l, ALT - 15 U/l, AST - 26 U/l, al. phosphatase - 415 U/l, LDH - 273 U/l, indicators of iron metabolism are within normal limits: serum Fe - 14.4 µm/l, THC - 69.9 µm/l. A negative result of the direct Coombs test allowed us to exclude autoimmune hemolytic anemia.
In hereditary microspherocytosis, the blood picture is characterized by a pathological triad on the part of erythrocytes: 1) microspherocytosis; 2) reticulocytosis; 3) reduced osmatic resistance. In our case, all three factors are well expressed. The patient was diagnosed with hereditary microspherocytic hemolytic anemia.
The child was discharged in stable condition on 06/09/09 with recommendations: observation by a hematologist and pediatrician, general and biochemical blood tests once every 3 months.
On June 24, 2009, patient S. was re-admitted to the oncohematology department. Upon admission, the condition was serious, complaints of lethargy, lack of appetite, poor sleep, fever up to 39.6°C, pallor, yellowness of the skin, dark urine. The pharynx is hyperemic and loose. The lymph nodes of the cervical group are enlarged. The abdomen is enlarged in volume, the liver protrudes from under the edge of the costal arch by 3.5 cm, the spleen by 5 cm.
General blood test dated June 24, 2009: HGB - 89 g/l, RBC - 3.4×10¹²/l, WBC - 27.7×109/l, Ht - 21%, MCV - 68.6 fl, MCH -25.5 pg, MSHC - 381 g/l, RDW - 20.8%, PLT - 260×109/l, ESR - 12 mm/h, in the formula: eosinophils - 4%, basophils - 1%, p/ I neutrophils - 4%, s/I neutrophils - 15%, lymphocytes - 20%, monocytes - 1%, atypical mononuclear cells - 55%. Reticulocytosis - 46.7%. Blood smears contain up to 50% microspherocytes. Indirect bilirubin - 41 µmol/l, ALT - 19 U/l, AST - 45 U/l, al. phosphatase - 316 U/l, LDH - 295 U/l. An increased content of stercobilin in the feces is determined. In the urine, urobilinuria is noted, the release of urates and uric acid crystals, explained by the increased breakdown of red blood cells. ELISA for Epstein-Barr virus (infectious mononucleosis) - positive.
Taking into account the child's intoxication, fever, enlarged cervical lymph nodes, high leukocytosis in the blood, mononuclear cells up to 55%, positive ELISA, infectious mononucleosis was diagnosed, which in turn was the trigger for the development of a hemolytic crisis in the boy.
Treatment was carried out: diet, bed rest, infusion and antiviral therapy. As a result of therapy, the fever stopped, the size of the liver, spleen and lymph nodes decreased. General blood test dated 07/06/09: HGB - 104g/l, RBC - 3.9×10¹²/l, WBC - 10.7×109/l, Ht - 28%, MCV - 73.3 fl, MCH - 26.4 pg, MSHC - 360 g/l, RDW - 21.8%, PLT - 452×109/l, ESR - 2 mm/h, in the leukocyte formula: eosinophils - 5%, p/n neutrophils - 5% , neutrophils - 25%, lymphocytes - 47%, monocytes - 8%, atypical mononuclear cells - 10%. Reticulocytosis - 9.3%. The number of microspherocytes decreased to 23%. Indirect bilirubin - 18.8 µmol/l. Appetite and sleep normalized. In a stable condition, on July 09, 2009, the child was discharged home with recommendations: observation by a hematologist once every 3 months, a general blood test with reticulocytes and a biochemical blood test once every 3 months, monitoring by ultrasound of the abdominal cavity once a year.
Clinical case No. 2. Patient G., 12 years old, on September 22, 2009, was transferred to the oncohematology department of the Yoshkar-Ola Children's City Hospital from the infectious diseases hospital. Upon receipt of a complaint of nasal congestion, sore throat, general weakness, enlarged cervical and occipital lymph nodes, fever up to 39.2°C, icteric skin, hepatosplenomegaly with pronounced density of parenchymal organs. From the anamnesis it is known that the girl’s father suffers from Gilbert’s syndrome and since birth the girl has developed yellowness of the skin and sclera due to fever and ARVI.
General blood test dated September 23, 2009: HGB - 61g/l, RBC - 1.8×10¹²/l, WBC - 10.7×109/l, Ht - 16%, MCV - 85 fl, MCH - 32, 5 pg, MSHC - 382g/l, RDW - 20.3%, PLT - 162×109/l, ESR - 48 mm/h, in the leukocyte formula: myelocytes - 1%, metamyelocytes - 2%, p/n neutrophils - 29%, neutrophils - 9%, lymphocytes - 24%, monocytes - 4%, atypical mononuclear cells - 31%. Reticulocytosis - 82.5%. Blood smears contain a significant number of microspherocytes. Osmotic resistance of erythrocytes: min - 0.72%, max - 0.42%, a sharp decrease in resistance to hypotonic solutions of table salt is determined. Increased levels of urobilin in urine and stercobilin in feces. Biochemical blood test dated September 23, 2009: ALT - 71 U/l, AST - 78 U/l, LDH 1234 U/l, direct bilirubin - 5.68 µmol/l, indirect - 54.8 µmol/l. Direct Coombs test is negative.
Severe normochromic anemia, the detection of a significant number of microspherocytes in blood products, a pronounced reticulocyte crisis, a sharp decrease in the osmotic resistance of erythrocytes, hyperbilirubinemia, as well as the presence of similar symptoms in the father gave grounds to diagnose the girl with a hemolytic crisis of hereditary microspherocytic anemia, provoked by infectious mononucleosis (Epstein virus ELISA - Barr - positive). An examination by an infectious disease specialist confirms the diagnosis of infectious mononucleosis.
The patient was prescribed infusion, antibacterial and antiviral therapy, and vitamins. During treatment, the patient's condition stabilized. General blood test dated 10/12/09: HGB - 106g/l, RBC - 3.5×10¹²/l, WBC - 3.9×109/l, Ht - 29.3%, MCV - 83.8 fl, MCH - 30.4 pg, MCHC - 362 g/l, RDW - 217.8%, PLT - 170×109/l, ESR - 8 mm/h, in the formula: basophils - 1%, p/n neutrophils - 4 %, neutrophils - 62%, lymphocytes - 20%, monocytes - 4%, atypical mononuclear cells - 9%. Reticulocytosis - 13.1%. Biochemical blood test: ALT - 41 U/l, AST - 32 U/l, direct bilirubin - 2.75 µmol/l, indirect - 24.35 µmol/l. According to abdominal ultrasound, positive dynamics are noted in the form of a decrease in the size of the liver and spleen. The girl was discharged home on October 13, 2009 with recommendations: observation by a hematologist, infectious disease specialist and pediatrician, complete blood count with reticulocytes once every 3 months, monitoring by ultrasound of the abdominal cavity.
Both patients are being monitored by a hematologist.
The diagnosis of congenital microspherocytosis sometimes presents certain difficulties. The most typical manifestations of hemolysis - yellowness of the sclera and skin - are not always correctly assessed by the doctor. The clinical cases we described are direct confirmation of this. In both patients, icterus of the sclera and skin was determined from birth, but a detailed examination was not carried out on the patients before the development of a hemolytic crisis. The presence of similar symptoms in the girl’s father (Gilbert’s syndrome?) suggests that patient G. inherited the disease from her father. The heredity of patient S. is unknown.
In both cases, the provoking factor in the development of the hemolytic crisis was the Epstein-Barr virus. Children have suffered from viral infections before, but they did not cause the development of a hemolytic crisis. Perhaps not every, but only a certain group of viruses can cause crises. This question remains open, since based on the example of two cases it cannot be argued that the Epstein-Barr virus always provokes the development of a hemolytic crisis in patients with hereditary microspherocytosis.
E.V. Shirdanina, Z.S. Gordeeva
Yoshkar-Ola Children's City Hospital, Yoshkar-Ola
Shirdanina Ekaterina Valerievna – doctor of the clinical diagnostic laboratory
Literature:
1. Pogorelov V.M., Kozinets G.I., Kovaleva L.G. Laboratory and clinical diagnosis of anemia. - Moscow. Medical Information Agency, 2004. - pp. 136-137.
2. Kokolina V.F., Rumyantsev A.G. Practical guide to childhood diseases. Pediatric Hematology/Oncology. - Volume 4. - M.: Medpraktika-M, 2004. - P. 169-172.
3. Vorobyov A.I. Guide to Hematology. - Volume 2. - M.: Medicine, 1985. - P. 46-54.
4. Kishkun A.A. Guide to laboratory diagnostic methods. M.: GEOTAR-Media, 2007. - pp. 468-680.
5. Kassirsky I.A., Alekseev G.A. Clinical hematology. - M.: Medicine, 1970. - P. 235-242.
6. Lugovskaya S.A., Pochtar M.E. Hematological atlas. - M.: Triada, 2004. - 227 p.
7. Kolokolov G.R., Gerasina E.V., Ananyev O.L. et al. Analyzes. Complete reference book. - M.: Eksmo, 2008. - P. 497-499.
8. Maksimov V.A., Damidovich K.K., Fedorchuk A.N. and others. Rare diseases, clinical syndromes and symptoms of diseases of the digestive system. - M.: Adamant, 2007. - P. 128-129.
9. Rukavitsin O.A., Skvortsov S.V., Zenina M.N. Hematology. Atlas-directory. St. Petersburg: Detstvo-Press, 2009. - pp. 219-220.
10. Pogorelov V.M., Kozinets G.I., Dyagileva O.A. and others. Color atlas of cells of the blood system. - M.: Practical Medicine, 2007. - P. 98-99.
Diagnosis of Minkowski-Choffard anemia
Identifying the disease is usually not difficult. The doctor will listen to the patient’s complaints and examine him. It is imperative to clarify whether the person’s closest blood relatives suffered from anemia. In addition to an external examination of the skin and mucous membranes, the doctor palpates the abdomen to determine whether the patient’s liver and spleen are enlarged in size. An ultrasound of these organs is mandatory.
Without laboratory tests, it will not be possible to confirm the diagnosis, so the doctor directs the patient to undergo the following tests:
- Analysis of urine. It can contain bilirubin, protein and urobilin in high concentrations.
- Blood for biochemical analysis. In this case, it will be possible to detect a decrease in cholesterol levels, an increase in the level of indirect bilirubin and lactate dehydrogenase.
- Blood for clinical analysis. In this case, an increase in the level of reticulocytes will be detected, the red blood cells themselves decrease in size. In addition, ESR increases, and a decrease in platelet and leukocyte levels may be observed. The blood color index will be below normal.
To confirm the diagnosis, it will be necessary to study the proteins of the red blood cell membranes using two-dimensional electrophoresis.
It is imperative to carry out a differential diagnosis of Minkowski-Choffard anemia with autoimmune diseases. First of all, it is necessary to carefully study the family history, since Minkowski-Choffard anemia is most often inherited. In addition, sick children have certain changes in the structure of the skull. If the doctor remains in doubt, the patient is given a Coombs test. When this test gives a negative result, the diagnosis is confirmed.
Diagnostics
The diagnosis of hereditary spherocytosis is made on the basis of laboratory tests, clinical manifestations and conversation with the patient to determine the hereditary predisposition to the disease.
The diagnosis is confirmed if hemolytic anemia, morphological features of red blood cells and their reduced osmotic stability are detected.
With hereditary spherocytosis, red blood cells have a spherical shape, their diameter is reduced and their thickness is increased, while the average cell volume remains normal. Hemoglobin levels are usually close to normal. Changes in the number of leukocytes (the leukocyte formula is shifted to the left, the level of neutrophils is increased) and ESR (increased) are observed only during a crisis, while the platelet level is normal.
A characteristic sign of the disease is a decrease in the osmotic resistance (stability) of red blood cells. The diagnosis can be confirmed only if it decreases significantly. It happens that with obvious spherocytosis, osmotic resistance remains normal. In this case, additional laboratory testing is required after daily incubation of red blood cells.
In the case of hereditary spherocytosis, differential diagnosis is required. The disease should be distinguished from other types of anemia. As a rule, this is hemolytic disease of newborns, autoimmune hemolytic anemia, viral hepatitis.
Treatment of Minkowski-Choffard anemia
Depending on the severity of the disease, the methods of its medicinal correction will differ. During the period of remission of anemia, therapy is not carried out. During an exacerbation of the pathology, the patient must be hospitalized.
Treatment boils down to the following activities:
- When the hemoglobin level decreases to 70 g/l, the patient is transfused with red blood cells.
- If the level of bilirubin in the blood is high, the patient is given albumin.
- To remove intoxication from the body, the patient is given infusion therapy.
- Choleretic drugs can improve the patient’s well-being during a hemolytic crisis.
Severe Minkowski-Choffard anemia requires surgical intervention. To do this, the patient's spleen is removed. This will not make it possible to completely get rid of anemia, but the person’s condition improves, since the life cycle of red blood cells can be extended.
After removal of the spleen, the patient will no longer be at risk of hemolytic crises. However, this procedure is contraindicated under the age of 5 years, since splenectomy can be fatal in children. In addition, the absence of a spleen in the body contributes to a decrease in immunity. A person becomes susceptible to various infections.
Another surgical method for treating anemia is endovascular occlusion. This procedure is an alternative to splenectomy. During the procedure, the patient is given a drug that causes a splenic infarction. A certain part of it does not die, but continues to function, so the patient’s condition improves, but immunity does not decrease.
If the disease is mild and the patient receives high-quality and timely treatment, then the prognosis for life is favorable. Removing the spleen avoids crises. Without surgery, the average duration of remission between hemolytic crises is 2 years.
Treatment
Treatment of this disease depends on the frequency of crises, clinical picture and age.
During a hemolytic crisis, conservative treatment with hospitalization is indicated. Therapy is aimed at combating hypoxia, hyperbilirubinemia, cerebral edema, hyperglycemic and other disorders that develop during a crisis. Treatment usually occurs according to the following scheme:
- For severe anemia, red blood cell transfusions are prescribed.
- After the crisis is over, choleretic drugs are prescribed, the diet and daily routine are expanded.
The only effective method of combating spherocytosis is removal of the spleen (splenectomy)
The operation is indicated for frequent crises, severe anemia, hepatic colic, and splenic infarction. Sometimes, along with this operation, removal of the gallbladder (cholecystectomy) is also performed. Before surgery, in case of severe anemia, red blood cell transfusion is performed.
As a rule, after surgery, recovery occurs in 100% of cases. At the same time, the red blood cells remain pathologically altered, that is, they have a spherical shape and low resistance to sodium chloride solutions. The cessation of the breakdown of red cells is due to the fact that there is no organ in which this destruction occurs.
In some cases, surgery must be performed immediately; even pregnancy is not a contraindication. Otherwise, cholelithiasis, liver cirrhosis, and hepatitis may develop.
The prognosis for hereditary spherocytosis is relatively good and people can live with this disease into old age. In severe cases of the disease, if help is not provided on time, death may occur.
If one of the parents is sick, the probability of developing pathology in children is less than 50%.
For microspherocytic anemia, a special diet is indicated. The patient's diet should contain foods containing large amounts of folic acid. Recommended to eat:
- porridge: buckwheat, millet, oatmeal;
- mushrooms;
- products made from wholemeal flour,
- beef liver;
- cheese;
- beans, soy;
- cottage cheese;
- green onions, cauliflower, chopped carrots.
Complications of Minkowski-Choffard anemia
In childhood, Minkowski-Choffard anemia can cause mental and physical development delays. Most often this is observed when the child does not receive treatment, or it is started too late.
In adulthood, the most common complication of Minkowski-Choffard anemia is gallstone disease, which develops against the background of increased levels of bilirubin in the blood. Often, hemolytic crisis is confused with obstructive jaundice, so the patient is not given adequate therapy. If stones are found in the patient's gall bladder, it is removed along with the spleen.
Prevention measures
Minkowski-Choffard anemia is a disease that is inherited, so it is impossible to prevent its development. People suffering from this pathology need to be registered with a hematologist.
The probability of a child being born with hemolytic anemia in a sick mother is equal to 50%. Therefore, such children should be thoroughly examined from birth.
Author of the article:
Shutov Maxim Evgenievich |
Hematologist Education: Graduated from Kursk State Medical University in 2013 and received a diploma in General Medicine. After 2 years, he completed his residency in the specialty “Oncology”. In 2021, she completed postgraduate studies at the National Medical and Surgical Center named after N.I. Pirogov. Our authors