Detailed description of the study
Potassium is one of the main cations (positively charged ions) in the human body. The main part of it is located inside cells and only a small part is in the extracellular space, including blood plasma. Potassium supports the electrical activity of cells, participates in the processes of conducting nerve impulses, muscle contraction, regulating water balance and acid-base balance in the blood.
The concentration of potassium in plasma depends on its intake, redistribution, excretion from the body and is regulated by various substances. In this way, a balance is maintained between intra- and extracellular potassium protein in the cell membrane (sheath) - the potassium-sodium pump. Insulin increases the influx of ions into cells, while alpha adrenergic agonists increase their exit from it. Blood pH also affects the level of this cation. With an increase in acidity (acidosis), potassium is released into the plasma, and with a decrease in acidity (alkalosis), there is an influx into the cell.
The macronutrient enters the body with food: meat, fruits and vegetables. A lot of it is found in apricots, prunes, potatoes, and legumes. The daily norm for humans is 4-5 g. Potassium is absorbed mainly in the small intestine and enters the cells. It is filtered in the kidneys and reabsorbed in large quantities - it enters the blood again. The hormone aldosterone, which is produced by the adrenal glands, increases the excretion of macronutrients by the kidneys. Potassium is also excreted through the intestines and sweat glands.
The concentration of potassium in plasma is quite low and even small fluctuations can lead to disturbances in the body. A test for potassium in the blood plasma may show a decrease in its level (hypokalemia). It occurs due to insufficient intake of the macroelement from food, increased excretion in the urine (some kidney diseases), loss of the ion from intestinal contents (vomiting, diarrhea), and increased production of aldosterone. The main symptoms of hypokalemia are associated with impaired neuromuscular conduction. A person experiences fatigue, muscle weakness, decreased muscle tone, and heart rhythm disturbances.
Hyperkalemia occurs due to excess potassium intake into the body, for example, during intensive infusion of potassium-containing drugs. Also, an increase in the level of the cation in plasma can be observed with insufficient excretion by the kidneys and increased release from cells (destruction of red blood cells, disseminated internal coagulation syndrome and other reasons). Symptoms of hyperkalemia: weakness in the legs, muscle paresis, abdominal pain, numbness, heart rhythm disturbances with a decrease in contraction frequency, and in severe cases, cardiac arrest.
A biochemical blood test for potassium allows you to diagnose its excess or deficiency and take timely measures to correct disorders.
Potassium and its significance in a biochemical blood test (K)
Potassium (“K”) is an essential chemical element that creates the necessary conditions for various reactions to occur. It is found in the body in the form of cations - positively charged ions located inside the cell (in the intracellular fluid).
The content in the intercellular fluid is no more than 2% - this is exactly the potassium that is determined during biochemical analysis. A change in this indicator can be observed both in various physiological conditions and in pathology, and a decrease in the concentration in the blood - hypokalemia - is life-threatening and can have serious consequences.
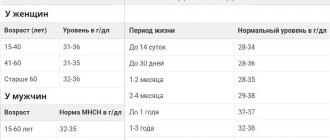
The norm is 3.4 – 5.0 mmol/l. At the same time, it provides:
- Correct heart rate;
- Myocardial contraction;
- The work of skeletal muscles;
- Conduction of nerve impulses through neurons;
- Water-salt balance.
For normal maintenance of the above functions, a sufficient supply of “K” from food is necessary, since there are no own reserves of this chemical element. A lot is found in vegetables (especially greens, potatoes), fruits and grains.
The level of “K” is affected by the rate of its elimination. A decrease in concentration is caused by:
- Kidney diseases;
- Water-salt and acid-base imbalance;
- Hormonal changes (especially mineralocorticoids);
- Uncontrolled use of diuretics;
- Intoxication with repeated vomiting or diarrhea;
- Severe inflammatory processes;
- Deficiency of other minerals;
- Disorders of respiratory and circulatory function.
Hypokalemia – level in biochemistry less than 3.5 mmol/l. At the same time, it develops:
- Severe fatigue;
- Muscle pain;
- Arrhythmia up to cardiac arrest;
- Reduced activity of gastrointestinal motility with subsequent constipation up to dynamic intestinal obstruction;
- Slowing reflexes;
- Slowing down of mental processes;
- Pathology of urination.
Conditions in which the norm is exceeded - hyperkalemia - are somewhat less common. However, this happens when:
- Kidney failure;
- Reverse mineralocorticoid disorders;
- Release of the cation into the intercellular space due to extensive injuries, tumors, poisoning, taking antibiotics or chemotherapy drugs, acid-base imbalance;
- Overdose of drugs with “K”;
- Enhanced diet.
The clinical picture of hyperkalemia is similar to the symptoms that occur with ion deficiency. This is due to the general nature of the occurrence of such signs - a violation of the electrochemical potential of membranes, which is characterized by:
- Arrhythmia;
- Weakening of skeletal muscles up to paralysis;
- Apathetic mood;
- Blood pressure instability (jumps from low to high).
Thus, the exchange of “K” is extremely important for maintaining the normal functioning of both each individual cell and the entire organism as a whole. Therefore, if any symptoms appear that may correspond to hypo- or hyperkalemia, it is imperative to take biochemistry tests.
1.General information
Potassium is one of the key macroelements in the human body. This substance, especially in interaction with sodium, is necessary for the regulation of contractile activity of muscles (including the myocardium), water-salt, acid-base and energy balance, higher nervous activity, normal fermentation and metabolism, secretory activity of the endocrine glands, functioning liver and kidneys. Up to half of all salts in the body are potassium compounds, and most of them are contained in intracellular liquid spaces.
For all micro- and macroelements, including potassium, a certain concentration range must be observed.
Otherwise, both deficiency and excess lead to the development of severe, sometimes life-threatening conditions. Hyperkalemia, i.e. Excessive potassium levels are a typical example.
A must read! Help with treatment and hospitalization!
How to lower potassium levels
If the amount of microelement is not controlled, the risk of a heart attack will increase. The first step in dealing with hyperkalemia should be to see a doctor. The specialist must check all medications and dietary supplements that the patient takes and find analogues to drugs that provoke an increase in potassium levels.
If total potassium is slightly increased, the problem can be dealt with without the help of a doctor. It is enough to refuse products with a high content of microelements in their composition. In particular, it is better to replace cow's milk with a natural oat or rice drink. Tomatoes and oranges are other common foods that increase potassium levels in the blood. As an alternative, nutritionists suggest eating grapes, apples or cranberries. You can also use less table salt.
You can get tested quickly and painlessly in our clinic. The technician will take a small amount of blood from a vein. The whole procedure will not take even ten minutes.
Important! During blood sampling for total potassium, it is strictly forbidden to clench your fist. This can lead to pseudohyperkalemia: the release of electrolyte outside the cells, the formation of a blood clot in a test tube. As a result, the analysis results will artificially increase by 1.5-2 mmol/l. At a rate of 3.5 – 5.0 mmol/l, such a “deviation” can be critical.
If your total potassium test results are outside the normal range, it does not necessarily indicate a medical condition that requires immediate medical attention. Some dietary supplements, multivitamins, and even herbs can increase potassium levels in the blood. The clinic specialists will definitely issue a sheet with the results of the analysis. But the indicators must be deciphered by a specialized doctor.
3. Symptoms and diagnosis
The clinical picture of hyperkalemia is not specific enough, so preliminary, presumptive diagnosis requires a doctor to have a lot of experience, attention and vigilance in this regard. Most often, changes in the ECG in combination with patient complaints of tachycardia (accelerated heartbeat), general malaise and constant weakness cause alarm. In more severe cases, cardiac abnormalities caused by excess potassium may take the form of arrhythmia, asystole, ventricular fibrillation, and ultimately cardiac arrest.
The diagnosis is established by comparing the existing clinical manifestations, the electrocardiographic picture and the results of laboratory tests (determining the concentration of potassium in the blood plasma). Depending on the most likely causes, an additional specialized examination is prescribed - for example, nephrological or endocrinological.
About our clinic Chistye Prudy metro station Medintercom page!
Interference:
- Beta blockers (rare), amiloride, aminocaproic acid, ACE inhibitors, cyclophosphamide, vincristine, arginine, cephaloridine, cyclosporine, digoxin, adrenaline (initial effect), foscarnet sodium, heparin, histamine (iv), isoniazid, lithium, mannitol.
- Beta-adrenergic agonists (salbutamol, terbutaline), albuterol, aminoglycosides, p-aminosalicylate, aminosalicylic acid (rare), amphotericin, azlocillin, bisacodyl, capreomycin, carbenicillin, carbenoxolone, cholestyramine, cisplatin, clopamide, corticosteroids, corticotropin, cyanocobal amine, dextrose anhydride, EDTA, diclofenamide, diuretics, enoxolone, fluconazole, glucagon, glucose, ifosfamide, insulin, levodopa, licorice, mezlocillin, nafcillin, penicillin G (sodium salt), phenolphthalein, piperacillin, polymyxin B, salicylates, sodium bicarbonate, sodium chloride, ticarzi llyn, theophylline.
2. Reasons
It would be logical to assume that hyperkalemia is caused by excessive consumption of potassium-rich foods, for example, dried fruits, legumes, potatoes, bananas, etc. However, in reality this is extremely rare: the body’s daily need for potassium is quite large (up to 5 g in adults), and with normal metabolism the balance is easily restored through the excretion of potassium in the urine.
Thus, one of the main factors in the development of hypercalcemia is disorders of the kidneys (chronic renal failure, diabetic nephropathy, etc.), when potassium excretion becomes difficult and it begins to be retained in the body. This can be caused, in particular, by hypocorticism (Addison's disease) - chronic functional insufficiency of the adrenal cortex, in which the kidneys simply do not receive the appropriate hormonal “commands”.
Another group of reasons is associated with long-term use of medications that contain potassium or prevent its excretion.
The massive release of potassium from cellular fluids into the bloodstream is accompanied by severe polytrauma with crushing of large volumes of muscle tissue, drug overdose, and extensive burns.
The progressive accumulation of potassium in the body can also be caused by pathologically rapid and intense hemolysis (decomposition of red blood cells) in anemia and some other diseases.
Rarely, hereditary hyperkalemia is recorded - familial hyperkalemic periodic paralysis.
Visit our Therapy page
4.Treatment
With moderate and severe severity, hyperkalemia requires emergency intensive care due to the threat of cardiac arrest. One of the emergency measures is the intravenous administration of a special solution (calcium, insulin, glucose), but its effect is short-lived, and after relief of life-threatening symptoms, further therapy is required. Potassium-absorbing sorbents are prescribed (which ensures its excretion in feces); diuretics are used if renal function is preserved. Stop taking any medications that in one way or another affect the circulation of potassium in the body. Limit potassium intake from food. Since hyperkalemia is almost always secondary, a diagnostic examination (if the cause of the detected hyperkalemia is unknown) and treatment of the underlying disease are mandatory.