Diabetes mellitus is such a common disease that there is hardly a person who has not heard of it or does not know that diabetes is manifested by elevated levels of sugar (glucose) in the blood. But not everyone suspects how dangerous it is. The fact is that diabetes has been the leading cause of death in the world for many decades. And every year the situation not only does not improve, but only gets worse. So, if you look at the report of the World Health Organization, the numbers are simply terrifying: between 2000 and 2021, mortality from diabetes increased by 70% and in 2021 amounted to 1.5 million people. That is, this is more than the new coronavirus infection claimed in a year. Moreover, 85% of these were patients with type 2 diabetes mellitus. But many people take this disease as an almost “normal condition” that does not require special attention. In fact, it is long-term neglect of diabetes that leads to such sad consequences.
Can you “feel” type 2 diabetes?
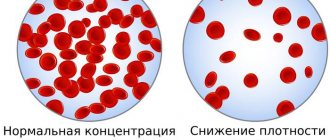
Unfortunately, slightly elevated blood sugar levels are not noticeable. This is the insidiousness of type 2 diabetes - a person feels good, but he already develops complications of diabetes. With higher glucose levels, dry mouth, thirst, and increased urination (especially at night) . But these symptoms are only a manifestation of the body’s protective reaction to elevated blood sugar levels and do not at all indicate complications of diabetes. By urinating more frequently, the body tries to get rid of excess glucose. Therefore, with normalization of blood sugar, these symptoms disappear very quickly. Chronic complications of diabetes, or also called “late complications,” arise due to damage to blood vessels and nerve endings, which are “sugared” by excess amounts of glucose. The very first manifestations of complications are: blurred vision, numbness of the toes, a feeling of “pins and needles” in the legs, and in men - impaired potency . In a later period, hemorrhages in the retina may occur, which often result in complete loss of vision, strokes, heart attacks , as well as gangrene of the lower extremities , requiring amputation of the legs.
Type 1 diabetes mellitus in adults
Diabetes mellitus is a major medical and social problem throughout the world. This is explained by its wide distribution, the severity of late complications, and the high cost of diagnostic and treatment tools that patients need throughout their lives.
According to experts from the World Health Organization, the total number of patients with all forms of diabetes mellitus today is over 160 million people. Every year, the number of newly diagnosed cases is 6–10% of the total number of patients, thus the number of people suffering from this disease doubles every 10–15 years. Type 1 diabetes is the most severe form of diabetes, accounting for no more than 10% of all cases of the disease. The highest incidence is observed in children aged 10 to 15 years - 40.0 cases per 100 thousand people.
An international expert committee, founded in 1995 with the support of the American Diabetes Association, proposed a new classification, which is accepted in most countries of the world as a recommendation document [10, 12]. The main idea underlying the modern classification of diabetes is a clear identification of the etiological factor in the development of diabetes [15].
Type 1 diabetes mellitus is a metabolic (metabolic) disease characterized by hyperglycemia, which is based on the destruction of β-cells, leading to an absolute deficiency of insulin. This form of diabetes was previously referred to as insulin-dependent diabetes mellitus or juvenile diabetes mellitus. The destruction of β-cells in most cases among the European population is of an autoimmune nature (with the participation of the cellular and humoral components of the immune system) and is caused by the congenital absence or loss of tolerance to β-cell autoantigens [8].
Multiple genetic predisposing factors lead to autoimmune destruction of β-cells. The disease has a clear association with the HLA system, with the DQ A1 and DQ B1 genes, as well as DR B1. HLA DR/DQ alleles can be both predisposing and protective [4].
Type 1 diabetes is often combined with other autoimmune diseases, such as Graves' disease (diffuse toxic goiter), autoimmune thyroiditis, Addison's disease, vitiligo and pernicytic anemia. Type 1 diabetes may be a component of an autoimmune syndrome complex (autoimmune polyglandular syndrome type 1 or 2, “rigid person” syndrome).
Summarizing the clinical and experimental data obtained to date, we can present the following concept of the pathogenesis of type 1 diabetes. Despite the appearance of an acute onset, type 1 diabetes develops gradually. The latent period can last for several years. Clinical symptoms appear only after 80% of β-cells have been destroyed. An autopsy study of pancreatic tissue from patients with type 1 diabetes reveals the phenomena of insulitis, a specific inflammation characterized by infiltration of islets with lymphocytes and monocytes.
The earliest stages of the preclinical period of type 1 diabetes are characterized by the appearance of clones of autoreactive T lymphocytes that produce cytokines, which leads to the destruction of β-cells. Insulin, glutamate decarboxylase, heat-shock protein 60, and fogrin are currently considered as putative primary autoantigens that, under certain conditions, cause proliferation of cytotoxic T-lymphocytes.
In response to the destruction of β-cells, plasma cells secrete autoantibodies to various β-cell antigens, which are not directly involved in the autoimmune reaction, but indicate the presence of an autoimmune process. These autoantibodies belong to the immunoglobulin G class and are considered as immunological markers of autoimmune damage to β-cells. There are islet cell autoantibodies (ICA - a set of autoantibodies to various cytoplasmic antigens of the β-cell), β-cell-specific autoantibodies to insulin, antibodies to glutamate decarboxylase (GAD), to phosphotyrosine phosphatase (IA-2), and fogrin. Autoantibodies to β-cell antigens are the most important markers of autoimmune destruction of β-cells and they appear in typical type 1 diabetes much earlier than the clinical picture of diabetes develops [4, 6]. Autoantibodies to islet cells appear in the serum 5–12 years before the first clinical manifestations of diabetes mellitus, their titer increases in the late stage of the preclinical period.
There are 6 stages in the development of type 1 diabetes, starting with genetic predisposition and ending with complete destruction of β-cells [1].
Stage 1 - genetic predisposition - is characterized by the presence or absence of genes associated with type 1 diabetes. The first stage occurs in less than half of genetically identical twins and in 2–5% of siblings. The presence of HLA antigens, especially class II - DR 3, DR 4 and DQ, is of great importance.
Stage 2 - the beginning of the autoimmune process. External factors that can play the role of a trigger in the development of autoimmune damage to β-cells can be: viruses (Coxsackie B virus, rubella, mumps, cytomegalovirus, Epstein-Barr virus), medications, stress factors, nutritional factors (use of infant formula containing animal proteins; products containing nitrosamines). The fact of exposure to various environmental factors can be established in 60% of patients with newly diagnosed type 1 diabetes.
Stage 3 - development of immunological disorders. Specific autoantibodies to various β-cell structures can be detected in the blood: insulin autoantibodies (IAA), ICA, GAD, IA2 and IA2b. In stage 3, there is impaired β-cell function and, as a result of a decrease in β-cell mass, loss of the first phase of insulin secretion, which can be diagnosed by performing an intravenous glucose tolerance test.
Stage 4 - severe immunological disorders - is characterized by impaired glucose tolerance, but there are no clinical signs of diabetes mellitus. When performing an oral glucose tolerance test (OGTT), an increase in glucose levels on an empty stomach and/or 2 hours after the OGTT is detected.
At stage 5, clinical manifestation of the disease is noted, since by this moment the bulk of β-cells (more than 80%) die. Residual low secretion of C-peptide persists for many years and is the most important factor in maintaining metabolic homeostasis. Clinical manifestations of the disease reflect the degree of insulin deficiency.
Stage 6 is characterized by a complete loss of functional activity of β-cells and a decrease in their number. This stage is diagnosed when there is a high level of glycemia, a low level of C-peptide and no response during the exercise test. This stage is called “total” diabetes. Due to the final destruction of β-cells at this stage, a decrease in the titer of antibodies to islet cells or their complete disappearance is sometimes observed.
There is also idiopathic type 1 diabetes mellitus, in which there is a decrease in β-cell function with the development of symptoms of insulinopenia, including ketosis and ketoacidosis, but there are no immunological markers of autoimmune destruction of β-cells. This subtype of diabetes mellitus occurs mainly among patients of African or Asian race. This form of diabetes mellitus has a clear inheritance. The absolute need for replacement therapy in such patients may appear and disappear over time.
As population studies have shown, type 1 diabetes among the adult population is much more common than previously thought. In 60% of cases, type 1 diabetes develops after age 20. The onset of diabetes in adults can have a different clinical picture. The literature describes the asymptomatic development of type 1 diabetes [13] in first- and second-degree relatives of patients with type 1 diabetes with a positive titer of autoantibodies to β-cell antigens, when the diagnosis of diabetes mellitus was made only based on the results of an oral glucose tolerance test.
The classic course of type 1 diabetes with the development of a state of ketoacidosis at the onset of the disease also occurs in adults [6]. The development of type 1 diabetes has been described in all age groups, up to the ninth decade of life [12].
In typical cases, the onset of type 1 diabetes has pronounced clinical symptoms, reflecting a deficiency of insulin in the body. The main clinical symptoms are: dry mouth, thirst, frequent urination, weight loss. Quite often the onset of the disease is so acute that patients can pinpoint the month, and sometimes even the day, when they first experienced the above symptoms. Rapid, sometimes up to 10–15 kg per month, loss of body weight for no apparent reason is also one of the main symptoms of type 1 diabetes. In some cases, the onset of the disease is preceded by a severe viral infection (influenza, mumps, etc.) or stress. Patients complain of severe weakness and fatigue. Autoimmune diabetes mellitus usually begins in children and adolescents, but can develop at any age.
If symptoms of diabetes mellitus are present, laboratory tests are necessary to confirm the clinical diagnosis. The main biochemical signs of type 1 diabetes are: hyperglycemia (as a rule, a high percentage of sugar in the blood is determined), glucosuria, ketonuria (the presence of acetone in the urine). In severe cases, decompensation of carbohydrate metabolism leads to the development of diabetic ketoacidotic coma.
Diagnostic criteria for diabetes mellitus:
- fasting plasma glucose more than 7.0 mmol/l (126 mg%);
- fasting capillary blood glucose more than 6.1 mmol/l (110 mg%);
- plasma glucose (capillary blood) 2 hours after a meal (or a load of 75 g of glucose) more than 11.1 mmol/l (200 mg%).
Determination of the level of C-peptide in serum allows one to assess the functional state of β-cells and, in doubtful cases, to distinguish type 1 diabetes from type 2 diabetes. Measuring C-peptide levels is more informative than insulin levels. In some patients at the onset of type 1 diabetes, a normal basal level of C-peptide may be observed, but there is no increase in it during stimulation tests, which confirms the insufficient secretory ability of β-cells. The main markers confirming the autoimmune destruction of β-cells are autoantibodies to β-cell antigens: autoantibodies to GAD, ICA, insulin. Autoantibodies to islet cells are present in the serum of 80–95% of patients with newly diagnosed type 1 diabetes and in 60–87% of individuals in the preclinical period of the disease.
The progression of β-cell destruction in autoimmune diabetes mellitus (type 1 diabetes) can vary [7].
In childhood, the loss of β-cells occurs rapidly and by the end of the first year of the disease the residual function fades away. In children and adolescents, the clinical manifestation of the disease usually occurs with symptoms of ketoacidosis. However, in adults there is also a slowly progressive form of type 1 diabetes mellitus, described in the literature as slowly progressive autoimmune diabetes of adults - Latent Autoimmune Diabetes in Adults (LADA).
Slowly progressive autoimmune diabetes of adults (LADA)
This is a special variant of the development of type 1 diabetes mellitus observed in adults. The clinical picture of type 2 diabetes and LADA at the onset of the disease is similar: compensation of carbohydrate metabolism is achieved through diet and/or the use of oral hypoglycemic drugs, but then, during a period that can last from 6 months to 6 years, decompensation of carbohydrate metabolism is observed and insulin need develops [18 , 19]. A comprehensive examination of such patients reveals genetic and immunological markers characteristic of type 1 diabetes.
LADA is characterized by the following features:
- age of debut, usually exceeding 25 years;
- clinical picture of type 2 diabetes without obesity;
- initially, satisfactory metabolic control achieved through the use of diet and oral hypoglycemic drugs;
- development of insulin requirements in the period from 6 months to 10 years (on average from 6 months to 6 years);
- presence of markers of type 1 diabetes: low level of C-peptide; the presence of autoantibodies to β-cell antigens (ICA and/or GAD); presence of HLA alleles at high risk of developing type 1 diabetes.
As a rule, patients with LADA do not have a clear clinical picture of the onset of type I diabetes, which is typical for children and adolescents. At onset, LADA is “masked” and initially classified as type 2 diabetes because the process of autoimmune β-cell destruction in adults may be slower than in children. Symptoms of the disease are erased, there is no pronounced polydipsia, polyuria, weight loss and ketoacidosis. Excess body weight also does not exclude the possibility of developing LADA. The function of β-cells fades slowly, sometimes over several years, which prevents the development of ketoacidosis and explains the satisfactory compensation of carbohydrate metabolism when taking PSSP in the first years of the disease. In such cases, type 2 diabetes is mistakenly diagnosed. The gradual nature of the development of the disease leads to the fact that patients seek medical help too late, having time to adapt to the developing decompensation of carbohydrate metabolism. In some cases, patients come to the doctor 1–1.5 years after the manifestation of the disease. In this case, all the signs of a sharp insulin deficiency are revealed: low body weight, high glycemia, lack of effect from PSSP. PZ Zimmet (1999) gave the following definition of this subtype of type 1 diabetes: “Autoimmune diabetes that develops in adults may not be clinically different from type 2 diabetes, and is manifested by a slow deterioration of metabolic control with the subsequent development of insulin dependence” [23]. Moreover, the presence in patients of the main immunological markers of type 1 diabetes—autoantibodies to β-cell antigens, along with low basal and stimulated levels of C-peptide—allows a diagnosis of slowly progressing autoimmune diabetes of adults [21].
Main diagnostic criteria for LADA:
- presence of autoantibodies to GAD and/or ICA;
- low basal and stimulated C-peptide levels;
- presence of HLA alleles at high risk for type 1 diabetes.
The presence of autoantibodies to β-cell antigens in patients with a clinical picture of type II diabetes at the onset of the disease has a high prognostic value regarding the development of insulin requirement [20, 22]. The results of the UK Prospective Diabetes Study (UKPDS), which examined 3672 patients with an initial diagnosis of type 2 diabetes, showed that antibodies to ICA and GAD have the greatest prognostic value in young patients (Table 1).
According to P. Zimmet, the prevalence of LADA is about 10–15% among all patients with diabetes mellitus and about 50% of cases occur in type 2 diabetes without obesity.
The results of our study showed [3] that patients aged 30 to 64 years, who at the onset of the disease had a clinical picture of type 2 diabetes without obesity, a significant decrease in body weight (15.5 ± 9.1 kg) and concomitant autoimmune thyroid diseases glands (DTZ or AIT), represent a group at increased risk of developing LADA. Determination of autoantibodies to GAD, ICA and insulin in this category of patients is necessary for timely diagnosis of LADA. Most often in LADA, antibodies to GAD are detected (according to our data, in 65.1% of patients with LADA), compared with antibodies to ICA (in 23.3% of LADA) and to insulin (in 4.6% of patients). The presence of a combination of antibodies is not typical. The titer of antibodies to GAD in patients with LADA is lower than in patients with type 1 diabetes with the same duration of the disease.
LADA patients represent a high-risk group for developing insulin requirements and require timely administration of insulin therapy. The OGTT results indicate the absence of stimulated insulin secretion in 46% of LADA patients and its decrease in 30.7% of patients already in the first 5 years of the disease. As a result of our study, 41.9% of patients with LADA, whose disease duration was no more than 5 years, were switched to insulin on average 25.2±20.1 months from the onset of the disease. This figure was significantly higher than in the group of patients with type 2 diabetes with the same duration of the disease (14% after 24±21.07 months from the onset of the disease, p < 0.05).
However, patients with LADA represent a heterogeneous group of patients. 53.7% of LADA patients have peripheral insulin resistance, while 30.7% of patients have a combination of insulin resistance and insulin deficiency due to autoimmune damage to β-cells [11, 16].
When choosing treatment tactics in patients with LADA, insulin secretion and peripheral tissue sensitivity to insulin should be assessed. A basal C-peptide level of less than 1 ng/ml (as determined by radioimmunoassay) indicates insulin deficiency. However, for patients with LADA, the absence of stimulated insulin secretion is more typical, while fasting insulin and C-peptide values are within normal limits (close to the lower limit of normal). The ratio of the maximum insulin concentration (at the 90th minute of the OGTT test) to the initial one is less than 2.8 with low initial values (4.6±0.6 µU/ml), which indicates insufficient stimulated insulin secretion and indicates the need for early administration insulin.
The absence of obesity, decompensation of carbohydrate metabolism when taking PSSP, low basal levels of insulin and C-peptide in LADA patients indicate a high probability of absence of stimulated insulin secretion and the need for insulin administration [6, 14].
If patients with LADA have a high degree of insulin resistance and insulin hypersecretion in the first years of the disease, it is recommended to prescribe drugs that do not deplete the function of β-cells, but improve the peripheral sensitivity of tissues to insulin, for example biguanides or glitazones (actos, avandia). Such patients are usually overweight and have satisfactory compensation of carbohydrate metabolism, but require further monitoring. To assess peripheral insulin resistance, the insulin resistance index can be used - Homa-IR = ins0/22.5 eLnglu0 (where ins0 is the fasting insulin level and glu0 is fasting plasma glucose) and/or the index of overall tissue sensitivity to insulin (ISI - insulin sensitivity index, or Matsuda index*), obtained based on the results of the OGTT. With normal glucose tolerance, Homa-IR is 1.21–1.45 points; in patients with type 2 diabetes, the Homa-IR value increases to 6 and even 12 points. The Matsuda index in the group with normal glucose tolerance is 7.3±0.1 UL–1 x ml x mg–1 x ml, and in the presence of insulin resistance its values decrease.
Preserving one's own residual insulin secretion in patients with type 1 diabetes mellitus is very important, since it is noted that in these cases the disease is more stable, and chronic complications develop more slowly and later. The importance of C-peptide in the development of late complications of diabetes mellitus is being discussed [6]. It was found that in the experiment, C-peptide improves kidney function and glucose utilization. It was found that infusion of small doses of biosynthetic C-peptide can affect microcirculation in human muscle tissue and renal function.
To determine LADA, more widespread immunological studies are indicated among patients with type 1 diabetes, especially in the absence of obesity and early ineffectiveness of PSSP. The main diagnostic method is the determination of autoantibodies to GAD and to ICA.
A special group of patients who also require close attention and where there is a need to determine autoantibodies to GAD and ICA are women with gestational diabetes mellitus (GDM). It has been established that 2% of women with gestational diabetes mellitus develop type 1 diabetes within 15 years. The etiopathogenetic mechanisms of the development of GDM are very heterogeneous, and for the doctor there is always a dilemma: is GDM the initial manifestation of type 1 or type 2 diabetes. McEvoy et al. published data on the high incidence of autoantibodies to ICA among Native and African-American women in America. According to other data, the prevalence of autoantibodies to ICA and GAD was 2.9 and 5%, respectively, among Finnish women with a history of GDM. Thus, patients with GDM may experience a slow development of insulin-dependent diabetes mellitus, as with LADA diabetes. Screening patients with GDM to determine autoantibodies to GAD and ICA makes it possible to identify patients who require insulin administration, which will make it possible to achieve optimal compensation of carbohydrate metabolism.
Taking into account the etiopathogenetic mechanisms of the development of LADA, the need for insulin therapy in these patients becomes obvious [14, 17], while early insulin therapy is aimed not only at compensating carbohydrate metabolism, but also allows maintaining basal insulin secretion at a satisfactory level for a long period. The use of sulfonylurea derivatives in LADA patients entails an increased load on β-cells and their faster depletion, while treatment should be aimed at maintaining residual insulin secretion and attenuating the autoimmune destruction of β-cells. In this regard, the use of secretogens in patients with LADA is pathogenetically unjustified.
After clinical manifestation, most patients with a typical clinical picture of type 1 diabetes in a period of 1 to 6 months experience a transient decrease in insulin requirements associated with an improvement in the function of the remaining β-cells. This is the period of clinical remission of the disease, or “honeymoon”. The need for exogenous insulin is significantly reduced (less than 0.4 units/kg body weight); in rare cases, even complete withdrawal of insulin is possible. The development of remission is a distinctive feature of the onset of type 1 diabetes and occurs in 18–62% of cases of newly diagnosed type 1 diabetes. The duration of remission ranges from several months to 3–4 years.
As the disease progresses, the need for exogenously administered insulin increases and averages 0.7–0.8 U/kg body weight. During puberty, the need for insulin can increase significantly - up to 1.0–2.0 U/kg body weight. With increasing duration of the disease due to chronic hyperglycemia, micro- (retinopathy, nephropathy, polyneuropathy) and macrovascular complications of diabetes mellitus (damage to coronary, cerebral and peripheral vessels) develop. The main cause of death is renal failure and complications of atherosclerosis.
Treatment of type 1 diabetes
The goal of therapy for type 1 diabetes is to achieve target values of glycemia, blood pressure and blood lipid levels (Table 2), which can significantly reduce the risk of developing micro- and marcovascular complications and improve the quality of life of patients.
The results of the multicenter randomized Diabetes Control and Complication Trail (DCCT) trial have convincingly shown that good glycemic control reduces the incidence of diabetes complications. Thus, a decrease in glycohemoglobin (HbA1c) from 9 to 7% led to a reduction in the risk of developing diabetic retinopathy by 76%, neuropathy by 60%, and microalbuminuria by 54%.
Treatment of type 1 diabetes includes three main components:
- diet therapy;
- physical exercise;
- insulin therapy;
- training and self-control.
Diet therapy and exercise
When treating type 1 diabetes, foods containing easily digestible carbohydrates (sugar, honey, sweet confectionery, sweet drinks, jam) should be excluded from the daily diet. It is necessary to control the consumption (count bread units) of the following products: grains, potatoes, corn, liquid dairy products, fruits. The daily caloric intake should be covered by 55–60% from carbohydrates, 15–20% from proteins and 20–25% from fats, while the proportion of saturated fatty acids should be no more than 10%.
The physical activity regime should be purely individual. It should be remembered that physical exercise increases tissue sensitivity to insulin, reduces glycemic levels and can lead to the development of hypoglycemia. The risk of hypoglycemia increases during exercise and for 12–40 hours after prolonged strenuous exercise. Light to moderate exercise lasting no more than 1 hour requires additional intake of easily digestible carbohydrates before and after exercise. With moderate long-term (more than 1 hour) and intense physical activity, adjustment of insulin doses is necessary. It is necessary to measure blood glucose levels before, during and after exercise.
Lifelong insulin replacement therapy is essential for the survival of patients with type 1 diabetes and plays a crucial role in the routine management of this disease. When prescribing insulin, different regimens can be used. Currently, it is customary to distinguish between traditional and intensified insulin therapy regimens.
The main feature of the traditional insulin therapy regimen is the lack of flexible adjustment of the dose of administered insulin to the glycemic level. In this case, self-monitoring of blood glucose is usually absent.
The results of multicenter DCCT convincingly proved the advantage of intensified insulin therapy in compensating carbohydrate metabolism in type 1 diabetes. Intensive insulin therapy includes the following:
- basal-bolus principle of insulin therapy (multiple injections);
- planned number of bread units for each meal (diet liberalization);
- self-monitoring (monitoring blood glucose throughout the day).
For the treatment of type 1 diabetes and the prevention of vascular complications, genetically engineered human insulins are the drugs of choice. Porcine and human semi-synthetic insulins obtained from pork are of lower quality compared to human genetically engineered ones.
Insulin therapy at this stage involves the use of insulins with different durations of action [2]. To create a basic insulin level, intermediate-acting or long-acting insulins are used (approximately 1 unit per hour, which is an average of 24-26 units per day). In order to regulate the level of glycemia after meals, short-acting or ultra-short-acting insulins are used in a dose of 1-2 units per 1 bread unit (Table 3).
Ultra-short-acting insulins (humalog, novorapid), as well as long-acting insulins (lantus) are insulin analogues. Insulin analogues are specially synthesized polypeptides that have the biological activity of insulin and have a number of specified properties. These are the most promising insulin preparations in terms of intensified insulin therapy. Insulin analogues Humalog (lispro, Lilly), as well as novorapid (aspart, Novo Nordisk) are highly effective in regulating postprandial glycemia. Their use also reduces the risk of hypoglycemia between meals. Lantus (insulin glargine, Aventis) is produced using recombinant DNA technology using a non-pathogenic laboratory strain of Escherichia coli (K12) as a producing organism and differs from human insulin in that the amino acid asparagine from position A21 is replaced by glycine and 2 molecules of arginine are added at C -end of the B-chain. These changes made it possible to obtain a peak-free, constant concentration profile of insulin action over 24 hours/day.
Ready-made mixtures of human insulins of various actions have been created, such as Mixtard (30/70), Insuman Comb (25/75, 30/70), etc., which are stable mixtures of short- and long-acting insulin in specified proportions.
To administer insulin, disposable insulin syringes are used (U-100 for administering insulin with a concentration of 100 U/ml and U-40 for insulin with a concentration of 40 U/ml), syringe pens (Novopen, Humapen, Optipen, Bd-pen, Plivapen) and insulin pumps. All children and adolescents with type 1 diabetes, as well as pregnant women with diabetes, patients with impaired vision and lower limb amputations due to diabetes should be provided with syringe pens.
Achieving target glycemic values is impossible without regular self-monitoring and adjustment of insulin doses. Patients with type 1 diabetes need to independently monitor glycemia daily, several times a day, for which not only glucometers can be used, but also test strips for visual determination of blood sugar (Glucochrome D, Betachek, Suprima Plus).
To reduce the incidence of micro- and macrovascular complications of diabetes, it is important to achieve and maintain normal levels of lipid metabolism and blood pressure [9].
The target blood pressure level for type 1 diabetes in the absence of proteinuria is blood pressure < 135/85 mmHg. Art., and in the presence of proteinuria - more than 1 g / day and in chronic renal failure - blood pressure < 125/75 mm Hg. Art.
The development and progression of cardiovascular diseases largely depends on the level of blood lipids. Thus, with cholesterol levels above 6.0 mol/l, LDL > 4.0 mmol/l, HDL < 1.0 mmol/ and triglycerides above 2.2 mmol/l, patients with type 1 diabetes have a high risk of developing cardiovascular disease complications. Therapeutic treatment goals that determine the low risk of developing cardiovascular complications in patients with type 1 diabetes are: total cholesterol < 4.8 mmol/l, LDL < 3.0 mmol/l, HDL > 1.2 mmol/l, triglycerides < 1.7 mmol/l.
In the coming decades, research will continue to create new pharmaceutical forms of insulin and means of their administration, which will make replacement therapy as close as possible to the physiological nature of insulin secretion. Research on islet cell transplantation is ongoing. However, a real alternative to allo- or xenotransplantation of cultures or “fresh” islet cells is the development of biotechnological methods: gene therapy, generation of β-cells from stem cells, differentiation of insulin-secreting cells from pancreatic duct cells or pancreatic cells [5]. However, today insulin still remains the main treatment for diabetes.
For questions regarding literature, please contact the editor.
I. V. Kononenko, Candidate of Medical Sciences O. M. Smirnova, Doctor of Medical Sciences, Professor Endocrinological Research Center of the Russian Academy of Medical Sciences, Moscow
*
Causes of type 2 diabetes mellitus
First of all, blood sugar levels are affected by what we eat and at what time, but especially in what quantity. Therefore, poor nutrition, of course, is one of the main causes of diabetes. This means... by correcting it, you can significantly reduce the risk of diabetes, and in some cases even prevent its development.
But, before we begin to describe the principles of nutrition, we need to figure out what other factors provoke the appearance of type 2 diabetes in order to understand who is at increased risk and who needs to pay special attention to their eating habits.
Risk factors for type 2 diabetes mellitus are:
- presence of close relatives with diabetes;
- overweight (BMI* from 25 to 29.9) or obesity (BMI more than 30). However, if you have an ideal body weight, but have a large waist (more than 80 cm in women, and more than 94 cm in men), this is also a risk factor for diabetes;
- increased glucose levels during pregnancy (gestational diabetes);
- elderly age;
- lack of physical activity;
- smoking.
*In medicine, the body mass index (BMI) is used to assess obesity, which is calculated using the formula: weight (kg)/height (m2).
Unfortunately, we cannot influence such risk factors as heredity and age, but other risks can be significantly reduced. And first of all, this applies to nutrition!
How can dietary changes help prevent diabetes?
When it comes to preventing diabetes, many people think about the need to almost completely limit carbohydrates in their diet. And at first glance, this looks quite logical - after all, it is carbohydrates that increase blood sugar levels. But still there is no need to rush into this. Firstly, carbohydrates are different, and secondly, they perform many useful functions in our body. You just need to understand what carbohydrates you can afford and at what time.
Carbohydrates differ from each other in the rate of absorption, that is, in the time they enter the blood. Therefore, they can be divided into “fast”, “medium” and “long” carbohydrates. “Fast” include foods and drinks to which sugar is added, including products that contain “natural sugars” (fructose) - honey, fruit juices, concentrates. They appear in the blood literally a few minutes after their consumption, sharply increasing blood sugar levels. These carbohydrates have the highest glycemic index*. “Medium” include : bread, cereals, pasta, potatoes, beets, carrots, corn. This kind of carbohydrates is absorbed much more slowly, and accordingly, glucose in the blood will also increase more slowly. But, if they are subjected to prolonged cooking, their absorption will be significantly accelerated. “Long” carbohydrates contain a large amount of plant fiber: tomatoes, cucumbers, zucchini, eggplant, cabbage, lettuce, and, of course, greens. And they are the most difficult for the body to process, so the sugar level will rise very slowly and evenly. Long carbohydrates have the lowest glycemic index.
*The glycemic index is a relative indicator that reflects the rate at which blood sugar levels rise after digesting certain foods.
Why us
- Doctors. Specialists of the highest qualification category, with academic degrees and extensive experience in practical and scientific work.
- Accurate diagnosis. Informative and painless examination: ultrasound of the abdominal organs, blood vessels, the whole range of necessary laboratory tests - determination of blood glucose levels and the amount of glycated hemoglobin, diagnosis of metabolic disorders and the presence of signs of atherosclerosis, etc.
- Complexity. Comprehensive treatment developed specifically for you, taking into account the full clinical picture with the participation of all necessary specialists: endocrinologist, nutritionist, therapist, cardiologist, ophthalmologist, etc.
- High tech. High-tech treatment using extracorporeal hemocorrection methods, which reduces the risk of developing microangiopathies (diabetic foot, visual impairment, renal failure) and makes it possible to increase tissue sensitivity to insulin and reduce the dosage of medications.
- Diabetes Check-up. The opportunity to be examined yourself and have your loved ones checked using the “Diabetes Check-up” program in order to eliminate even the slightest chance of developing this pathology in your loved ones.
- Convenience and comfort. All diagnostic and treatment units are located in a single complex of our hospital, which significantly saves the time and effort of our patients.
How does the time of day affect the absorption of carbohydrates?
Scientists have long noticed that the same product is absorbed differently by the body depending on the time of day. And as it turned out, this is due to the production of hormones, or rather, the predominance of one or another hormone. The fact is that hormones that affect carbohydrate metabolism are produced at a strictly defined time, that is, they have their own daily rhythm. At the same time, some hormones promote the absorption of carbohydrates, while others, on the contrary, suppress this process. And it doesn’t matter what time we wake up or go to bed - hormones do not adapt to us and continue to work in their own way. And, if our nutrition does not coincide with the biological rhythm, then the risk of diabetes increases significantly.
So how can you distribute carbohydrates throughout the day so that your blood sugar doesn't spike? You just need to follow a few simple rules:
- You can indulge in a lot at breakfast The fact is that in the morning, cells absorb glucose least of all due to low sensitivity to insulin. And the culprit is the stress hormone cortisol, which is produced in the early morning hours, helping the body adapt after sleep. But in the future, all sugar not absorbed by cells is converted into a useful supply of energy - glycogen. But! After consuming “medium” and “fast” carbohydrates, a feeling of hunger sets in very quickly. Therefore, if you add protein foods with a sufficient amount of fat to your diet, you will be provided with long-term satiety, and the coming day will allow you to use up all the “eaten” calories. However, for those who are planning a second breakfast (snack), of course, it is better not to overload the first meal with calories.
- At lunch , cortisol still continues to be produced, but in smaller quantities, so you can eat “medium” carbohydrates in the form of side dishes (stewed vegetables, potatoes, cereals, pasta). But it’s better not to eat sweets. Even during the daytime, “long” carbohydrates (salads) and protein foods (meat, fish, poultry) must be present.
- In the evening, insulin replaces cortisol. And all the “fast” and “medium” carbohydrates eaten begin to be instantly absorbed, predisposing to weight gain. Accordingly, the load on the body increases. Therefore, at dinner, it is better to give preference to “long” carbohydrates (fresh vegetables) and protein foods (cottage cheese, kefir, poultry, fish), but with a low fat content.
- Try to your main meal (in terms of calories) either for breakfast or lunch . Eating high-calorie foods in the evening increases the risk of developing diabetes, even in people with ideal body weight. But... a very early breakfast at 4-5 am, as well as a late dinner, can provoke the development of diabetes. In the early morning, the sleep hormone melatonin still continues to be produced, which suppresses insulin production, thereby increasing the risk of developing diabetes. Therefore, if you still have to have breakfast so early, it is better to give preference to dishes high in protein and fat (omelet, boiled eggs, cottage cheese, cheeses, avocado).
- And a little more about sweets: as mentioned earlier, it is preferable to eat “fast” carbohydrates in the morning and in small quantities , and, best of all, after eating foods that contain a large amount of plant fiber . Thanks to “long” carbohydrates, sweets will be absorbed more evenly and without a sharp increase in blood sugar.
- And, lastly, your daily diet should contain such an amount of calories that your body weight remains normal , since with every extra kilogram the risk of developing diabetes increases. If you are still overweight or obese, you should make every effort to lose weight.
Proper nutrition significantly reduces the risk of developing diabetes, but don’t forget about physical activity and quitting smoking!
Exercise and smoking cessation
According to Russian and foreign diabetic associations, very little is needed to prevent the development of diabetes - just 150 minutes of active physical activity per week. Of course, we are not talking about calm walking, but the type of activity does not matter. The main thing is that the classes give you pleasure (this will prevent you from giving up in the near future) and are regular.
And, if you smoke, quitting will further reduce your risk of developing diabetes. So far, the opinions of scientists about why smoking provokes diabetes are quite contradictory, but the very fact of the influence of smoking on the development of diabetes does not raise any doubts, even despite the fact that smokers, on average, have less body weight.
I hope that the information in this article will help you avoid developing type 2 diabetes. But in order not to miss the onset of the disease, I recommend checking the level of glucose (sugar) in the blood at least once a year. And for those who have risk factors for diabetes, in addition to glucose, it is advisable to check the level of insulin in the blood by calculating the HOMA-IR* index, since this indicator is the most sensitive and often increases long before the development of the disease.
*HOMA-IR index is an indicator that is used to assess the sensitivity of body cells to insulin and reflects the risk of developing type 2 diabetes.
You can find out your risk of developing diabetes and get a detailed consultation with an endocrinologist lasting 1 hour at the Expert medical center. Also in our center there is a “Check-up Diabetes Diagnostics” , which includes all the necessary studies and consultation with an endocrinologist.
Treatment of diabetes
- A decrease in the level of glycated hemoglobin by 1% reduces the likelihood of complications overall by 21%, myocardial infarction by 16%, and cerebral hemorrhage (stroke) by 44%.
- It is enough to undergo a course of vascular therapy 2 times a year to stop the development of complications of diabetes mellitus.
- The use of extracorporeal hemocorrection methods increases the bioavailability of prescribed medications, thereby reducing their dosage and increasing the effectiveness of therapy.
Therapy for this pathology should be comprehensive and pursue the achievement of individual goals set by the doctor depending on the length of the disease, age, existing complications or concomitant diseases, especially cardiovascular ones:
- Regular self-monitoring of glucose levels, which should correspond to the set goals
- Prevention and regression of blood vessel lesions.
- Restoring full blood supply to organs and tissues of the body.
- Prevention of complications.
At the Yauza Clinical Hospital, treatment of diabetes mellitus and its complications is carried out using carefully selected drug therapy (prescription of insulin or drugs that improve the uptake of glucose by cells, symptomatic treatment depending on the clinical picture) and regulation of the patient’s diet. And also with the help of innovative methods of extracorporeal hemocorrection (EG), which make it possible to prevent, stop the progression or even reduce the severity of vascular complications of diabetes - the main cause of its other consequences (pathologies of the heart, kidneys, eyes, feet).
Extracorporeal hemocorrection in the treatment of diabetes mellitus
EG allows:
- cleanse the plasma of substances that damage the vascular wall and nerve fibers in diabetes, and also reduce the concentration of these substances in tissues,
- reduce blood viscosity and its tendency to form blood clots,
- restore red blood cell membranes and their properties for delivering oxygen to tissues,
- activate blood microcirculation in tissues and internal organs, restoring their normal functioning,
- improve the impaired sensitivity of body tissues to insulin.
Two methods are used:
- Cryoapheresis: blood plasma is exposed to low temperatures in the presence of a certain substance (heparin), causing the appearance of cryoprecipitate, which includes a whole range of harmful substances; then it is removed, and the purified plasma is returned to the patient.
- Cascade plasma filtration: plasma passes through a filter that has a membrane with a certain pore size. This filter removes harmful substances from the plasma, and the purified plasma is returned to the patient's body.
After the first session of the chosen procedure, experts note an improvement in the state of health in most patients, and the course of treatment provides the following effects:
- The progression of diabetic changes in the retina, glomeruli of the kidneys, blood vessels of the heart, brain, lower extremities, and peripheral nerves is inhibited.
- The pain syndrome associated with ischemia and neuropathy loses its intensity or disappears altogether.
- Trophic ulcers heal or decrease in size.
- Blood glucose levels decrease.
- Signs of diabetic nephropathy become less pronounced, kidneys work better.
- Vision improves.
- Working capacity may be restored.
Extracorporeal hemocorrection is not a panacea, but this technique reduces the risk of developing complications such as diabetic foot, retino- and nephropathies, disruption of the nervous system, and patient disability. And comprehensive treatment of diabetes mellitus returns the patient the opportunity to do what he loves and lead an active lifestyle.