Symptoms of caffeine overdose
Unfortunately, drinking coffee has become a habitual ritual for many. In an attempt to get themselves into working condition as quickly as possible, people turn to coffee almost immediately after sleep. However, the negative consequences of such a habit will not be long in coming.
Often the symptoms of a caffeine overdose are perceived by people as negative consequences of daily stress, anxiety and an unsettled lifestyle:
- insomnia;
- depression;
- nervousness and irritability;
- increased anxiety.
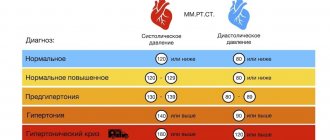
To a lesser extent, symptoms of caffeine overdose include frequent urination (enuresis), fluctuations in blood pressure, increased body temperature, heartburn, nausea, diarrhea, deterioration of the skin (caused by dehydration), vision problems and dizziness.
Caffeine-containing drinks are recommended to be consumed with caution by people with increased nervous excitability, cardiac arrhythmia, peptic ulcer, gastritis, atherosclerosis and glaucoma.
Rice. 1. Caffeine content in coffee and tea.
What to do if you overdose on caffeine
People who lead a hectic lifestyle and have health problems are more susceptible to symptoms of caffeine overdose. If you feel unwell (for example, with a sharp increase in blood pressure, high heart rate and dizziness) due to excessive consumption of caffeine-containing drinks, you must provide first aid and call a doctor.
To avoid possible death, you need to know what to do if you overdose on caffeine. We also advise you not to neglect calling a doctor, since at first glance, a slight deterioration in health may be a signal of a serious disruption in the functioning of the cardiovascular system.
Treatment of caffeine overdose
If possible, have someone nearby and follow the guidelines for treating caffeine overdose until doctors arrive:
- loosen your tie and/or belt;
- loosen tight clothing;
- open a window or go out into the fresh air;
- drink clean water until completely saturated;
- do not hold back your gag reflex.
Further treatment of caffeine overdose will be carried out by doctors. If the current state of health upon examination raises concerns among physicians, treatment of caffeine overdose will need to be performed in a medical facility. It may include gastric lavage, intravenous administration of magnesium sulfate, diprazine and morphine hydrochloride, as well as drugs to reduce agitation and normalize the functioning of internal organs. In severe cases, plasma substitutes are used to treat caffeine overdose.
Rice. 2. The ratio of patients with pathology of the cardiovascular system per 100 thousand population in the Russian Federation (according to Rosstat).
Caffeine prevents the development of atrial fibrillation
It is traditionally believed that patients with cardiac arrhythmias should not drink caffeine-containing drinks. The data obtained refuted this opinion. After a recent meta-analysis, scientists concluded that there is an inverse relationship between regular caffeine consumption and the risk of developing atrial fibrillation. Data published in the Canadian Journal of Cardiology report that, contrary to popular belief that caffeine can cause heart rhythm disturbances, this substance not only does not increase the risk of developing atrial fibrillation, but, on the contrary, helps reduce it. According to one of the researchers, Dr. Stanley Nuttel (University of Montreal), these data are comforting and reassuring for patients with atrial fibrillation who have to abstain from drinking coffee and other caffeinated drinks.
Caffeine and atrial fibrillation: feedback.
Caffeine is one of the main components of frequently consumed drinks such as coffee and cola. The purpose of the meta-analysis was not only to clarify the relationship between regular consumption of these drinks with the risk of developing atrial fibrillation, but also to investigate the dependence of the effect of caffeine consumption on its dose in such patients.
A meta-analysis by Cheng et al. , therefore, consisted of six prospective cohort studies that included a total of 228,465 participants. Three of these studies were conducted in the USA, two in Sweden and one in Denmark. The duration of the studies ranged from 4 to 25 years, and 4,261 participants developed atrial fibrillation during this time. The average age of individuals who took part in the studies was from 53 to 63 years. The prevalence of smoking among them ranged from 5.8% for participants with low caffeine intake to 76.8% for those with large amounts of caffeine-containing drinks.
In all studies, the incidence of atrial fibrillation was also documented in the form of electrocardiographic data. Three studies assessed daily caffeine intake by counting the frequency of drinking coffee or other caffeinated beverages over the past year, recalculating the average dose for each day. Three other studies documented exactly the daily dose of caffeine for each subject.
The pooled relative risk of incidence of atrial fibrillation among regular caffeine users was 0.90 (95% CI 0.81–1.01; p=0.07). There was significant heterogeneity among the three studies (I2=73%; p=0.002).
There was an inverse relationship between the development of cardiac arrhythmias and caffeine intake (p=0.015). The pooled results, taking into account the associated risks for developing atrial fibrillation, demonstrated a reduction in the incidence of atrial fibrillation by 11% with low-dose caffeine (relative risk 0.89, 95% CI 0.80–0.99; p=0.032) and 16% with high-dose caffeine (relative risk). risk 0.84, 95% CI 0.75–0.94; p=0.002). The overall incidence of atrial fibrillation showed a 6% reduction with daily consumption of 300 mg caffeine (relative risk 0.94, 95% CI 0.90–0.99; p=0.015)
Second cup?
In discussing their findings, the authors suggest that regular caffeine consumption may have a modest protective effect against the development of atrial fibrillation for a variety of reasons. One meta-analysis study demonstrated that 400 mg of caffeine did not cause changes in the P wave compared with pre-dose electrocardiogram data. Other studies have confirmed this, indicating that the same 400 mg of caffeine does not change such electrocardiographic parameters as the PR interval, the duration of the QRS complex, the QT interval, including the corrected one, and the RR intervals.
Other studies have demonstrated that 300 mg of caffeine may increase the incidence or severity of ventricular arrhythmias during the recovery phase after myocardial infarction. But even in patients with severe ventricular arrhythmias, caffeine does not significantly change the severity of this pathology.
The authors note that the lack of proarrhythmic ability of caffeine when taken regularly in drinks containing it is most likely due to the fact that the adrenergic effect of caffeine is weakened with daily consumption.
Another reason for the reduced risk of atrial fibrillation with caffeine consumption, according to the authors of the meta-analysis, is its antifibrotic effect. Heart rhythm disturbances occur when the composition of its tissues changes, in particular the replacement of cardiomyocytes with connective tissue, which leads to fibrosis.
Warning.
The study authors caution, however, that they believe that a patient with established atrial fibrillation should discuss the possibility of drinking caffeinated beverages with their healthcare provider. However, according to Dr. Nutella, the encouraging results obtained allow even patients with existing atrial fibrillation to drink one cup of coffee a day with a clear conscience.
Cheng M, Hu Z, Lu X, et al. Caffeine intake and atrial fibrillation incidence: Dose response meta-analysis of prospective cohort studies. Canadian J Cardiol 2014
Note!
Description of the drug Caffeine-sodium benzoate table. 200 mg No. 10 on this page is a simplified author’s version of the apteka911 website, created on the basis of the instructions for use.
Before purchasing or using the drug, you should consult your doctor and read the manufacturer's original instructions (attached to each package of the drug). Information about the drug is provided for informational purposes only and should not be used as a guide to self-medication. Only a doctor can decide to prescribe the drug, as well as determine the dose and methods of its use.
Pharmacological properties
Caffeine sodium benzoate is used primarily as a central nervous system stimulant, since caffeine enhances and regulates excitation processes in the cerebral cortex, enhances positive reflexes, and increases motor activity.
this effect is transformed into an increase in mental and physical performance, a decrease in feelings of fatigue and drowsiness. the effect of the drug largely depends on the type of higher nervous activity. In addition, the observed effects are dose-dependent, and caffeine in high doses can lead to depletion of nerve cells. the drug weakens the effect of sleeping pills and narcotics, increases reflex excitability of the spinal cord, and stimulates the respiratory and vasomotor centers. cardiac activity increases, myocardial contractions become more intense and more frequent. the hypertensive effect of caffeine sodium benzoate is observed only in shock and collapsed conditions. diuresis under the influence of the drug is slightly increased (mainly due to a decrease in the reabsorption of electrolytes in the renal tubules). The mechanism of action of the drug is due to the inhibition of the phosphodiesterase enzyme by caffeine, leading to the accumulation of cAMP inside cells. The latter enhances glycogenolysis, stimulates metabolism in organs and tissues, including the central nervous system and muscles. An important link in the mechanism of the stimulating effect of the drug is the binding of caffeine to purine receptors in the brain.
Caffeine sodium benzoate 100 mg No. 6 tablets
Compound
1 tablet contains caffeine (in the form of sodium benzoate) 0.1 or 0.2 g Packaging In a strip pack of 6 or 10 pcs.
pharmachologic effect
Analeptic, psychostimulant, cardiotonic. It has a stimulating effect on the central nervous system, increases reflex excitability of the spinal cord, stimulates the respiratory and vasomotor centers, regulates and enhances excitation processes in the cerebral cortex. Increases mental and physical performance, reduces fatigue and drowsiness. Increases the strength and frequency of heart contractions (especially in large doses), increases blood pressure during hypotension (does not change normal). Dilates the blood vessels of skeletal muscles, heart, kidneys, bronchi, narrows the abdominal organs. Increases the secretory activity of the stomach. Reduces platelet aggregation. Has a moderate diuretic effect. Blocks central and peripheral adenosine receptors. Stabilizes transmission at dopaminergic synapses, beta-adrenergic synapses of the hypothalamus and medulla oblongata, cholinergic synapses of the cortex and medulla oblongata, noradrenergic synapses. The effect of the drug largely depends on the type of nervous system in the patient, so the dosage of caffeine should take into account the individual characteristics of nervous activity.
indications for use
Diseases accompanied by depression of the functions of the central nervous system, cardiovascular system, respiratory system (drug poisoning, infectious diseases, etc.). For cerebral vascular spasms, to increase mental and physical performance, to eliminate drowsiness, breathing problems, enuresis in children.
Contraindications
Severe arterial hypertension, organic diseases of the cardiovascular system (including atherosclerosis), increased excitability, glaucoma, sleep disorders, old age. Side effects Anxiety, agitation, insomnia, tachycardia, arrhythmias, increased blood pressure, nausea, vomiting. With long-term use, slight addiction is possible (a decrease in the effect of caffeine is associated with the formation of new adenosine receptors in brain cells). Sudden cessation of caffeine administration may lead to increased central nervous system inhibition with symptoms of fatigue, drowsiness and depression.
Directions for use and doses
Tablets are taken orally - 0.025-0.1 g 2-3 times a day. The maximum single dose for adults is 0.4 g, daily dose is 1 g.
Side effects
From the nervous system: agitation, anxiety, tremor, restlessness, headache, dizziness, epileptic seizures, increased reflexes, tachypnea, insomnia; with sudden withdrawal - increased inhibition of the central nervous system, increased fatigue, drowsiness, muscle tension. From the cardiovascular system: palpitations, tachycardia, arrhythmias, increased blood pressure. From the digestive system: nausea, vomiting, exacerbation of peptic ulcer. Other: nasal congestion, with prolonged use - addiction, drug dependence.
special instructions
It should be borne in mind that sudden cessation of use may lead to increased inhibition of the central nervous system (drowsiness, depression). The effect on the central nervous system depends on the type of nervous system and can manifest itself as both excitation and inhibition of higher nervous activity. Due to the fact that the effect of caffeine on blood pressure consists of vascular and cardiac components, as a result, both the effect of stimulating the heart and inhibition (weak) of its activity can develop. For apnea in newborns and infants in the postoperative period (prevention), caffeine or caffeine citrate is used, but not caffeine sodium benzoate. Excessive consumption during pregnancy can lead to spontaneous abortions, slowing of intrauterine development of the fetus, arrhythmia in the fetus; There may be disturbances in skeletal development when using large doses and a slowdown in skeletal development when using lower doses. Passes into breast milk in small quantities, but accumulates in infants and can cause hyperactivity and insomnia. Do not take before bedtime.
Drug interactions
Reduces the effect of sleeping pills and narcotics, increases (improving bioavailability) - acetylsalicylic acid, paracetamol and other non-narcotic analgesics. Improves the absorption of ergotamine in the gastrointestinal tract.
Overdose
When abused, caffeine (more than 300 mg per day, i.e. four cups of natural coffee, 150 ml each) can cause anxiety, restlessness, tremors, headache, confusion, and cardiac extrasystoles. In newborns (including premature infants), at a blood plasma concentration of 50 mg/ml, toxic effects are possible: anxiety, tachypnea, tachycardia, tremor, increased Moro reflex, and at higher concentrations - convulsions.
Storage conditions
In a place protected from light.
Caffeine citrate 20 mg/ml 1 ml No. 10 ampoules
Content
Pharmacological properties Indications for use Contraindications With caution Use during pregnancy and breastfeeding Method of administration and dosage Dose adjustment and monitoring Side effects Overdose Interaction with other drugs Special instructions Storage conditions Expiration date
Pharmacological properties
Pharmacodynamics
The structure of caffeine is similar to methylxanthines - theophylline and theobromine. Most of its actions are associated with the blockade of adenosine receptor subtypes A1 and A2A. The main effect of caffeine is to stimulate the central nervous system. This underlies the activity of caffeine in apnea of prematurity, where it is thought to have the following effects:
- stimulation of the respiratory center;
- increase in minute ventilation;
- decreased sensitivity threshold to hypercapnia;
- increased response to hypercapnia;
- increased skeletal muscle tone;
- reducing diaphragm weakness;
- increase in basal metabolic rate;
- increased oxygen consumption.
Indications for use
Treatment of primary apnea (stopping breathing) in premature newborns.
Contraindications
Hypersensitivity to the active substance or any component of the drug.
Carefully
Concomitant diseases in the fetus of the cardiovascular system; the presence of convulsive syndrome, liver or kidney failure; excessive consumption by the mother of a newborn of caffeine-containing products in the period immediately before birth. Caffeine citrate should be administered with extreme caution to patients with gastroesophageal reflux.
Use during pregnancy and breastfeeding
Pregnancy
Animal studies have shown that high doses of caffeine have embryotoxic and teratogenic effects. These effects are not significant with short-term use of the drug in premature newborns.
Breast-feeding
Caffeine is excreted in breast milk and easily passes through the placenta into the fetal bloodstream. Breastfeeding mothers of newborns receiving caffeine citrate treatment should avoid foods, drinks, and medications that contain caffeine. In preterm neonates whose mothers consumed large amounts of caffeine before birth, baseline plasma caffeine concentrations should be determined before initiating treatment with caffeine citrate.
Fertility
The effects on the reproductive system observed in animals do not apply to premature neonates.
Directions for use and doses
Caffeine citrate can be prescribed as an intravenous infusion or orally. The drug cannot be prescribed intramuscularly, subcutaneously, intrathecally, or as an intraperitoneal injection.
If intravenous administration is used, caffeine citrate is administered as a controlled intravenous infusion using a syringe infusion pump or other metered infusion device. Caffeine citrate can be used either without dilution or in dilution with the following sterile preparations: glucose 50 mg/ml (5%), or sodium chloride 9 mg/ml (0.9%), or calcium gluconate 100 mg/ml (10%). ), immediately after removal from the ampoule.
The drug should only be used in a neonatology intensive care unit, which is equipped with appropriate equipment for patient management and monitoring.
Treatment with caffeine citrate should be initiated under the supervision of a physician experienced in the neonatal intensive care unit.
Recommended dosage regimen for treatment-naive infants: loading dose of caffeine citrate 20 mg per kg body weight by slow intravenous infusion over 30 minutes using a syringe infusion pump or other metered infusion device. After a 24-hour interval, a maintenance dose of 5 mg per kg body weight is prescribed by slow intravenous infusion over 10 minutes; the infusion is repeated every 24 hours. Alternatively, a maintenance dose of 5 mg per kg body weight can be given orally by nasogastric tube every 24 hours.
Recommended loading and maintenance doses of caffeine citrate are given in the following table, which shows the correspondence between the injection volume and the administered dose of caffeine citrate. The dose, expressed as caffeine base, is half the dose of caffeine citrate (20 mg caffeine citrate is equivalent to 10 mg caffeine base).
Premature neonates with insufficient therapeutic response to the recommended loading dose may be given a repeat loading dose of a maximum of 10–20 mg/kg after 24 hours.
Higher loading doses of 10 mg/kg body weight should only be administered if no improvement is observed, taking into account the possibility of accumulation of caffeine due to its longer T1/2 in preterm neonates and the progressively increasing capacity to metabolize caffeine depending on from age. Plasma concentrations of caffeine should be monitored as clinically indicated. The diagnosis of apnea in preterm infants should be reconsidered if patients do not respond sufficiently to a second loading dose or to maintenance doses of 10 mg/kg/day.
Dose adjustment and monitoring
It is advisable to periodically monitor plasma caffeine concentrations during treatment in cases where no improvement is observed or signs of toxicity appear. Dosage adjustments may be necessary after monitoring plasma caffeine concentrations in high-risk situations such as:
- very premature infants (less than 28 weeks of gestation and/or weighing less than 1 kg), especially those receiving parenteral nutrition;
- children with liver and kidney failure;
- children with seizure disorders;
- children with proven and clinically significant heart pathology;
- children concomitantly receiving medications that may affect the metabolism of caffeine;
- children whose mothers consume caffeine during breastfeeding.
Baseline caffeine concentrations should be determined in the following patients:
- children whose mothers may have consumed large amounts of caffeine before they were born;
- children previously treated with theophylline.
Duration of treatment
The optimal duration of treatment has not been established; the average treatment period is 37 days. In clinical practice, treatment is continued until the child reaches postmenstrual age 37 weeks (gestational age + calendar age, i.e. number of weeks of pregnancy + actual age after birth in weeks), by this period attacks of apnea of prematurity resolve spontaneously. However, this limit may be revised depending on the clinical situation, in individual cases depending on the response to treatment, the continuation of apnea episodes despite treatment, or other clinical considerations.
It is recommended to discontinue use of caffeine citrate if the patient does not experience significant apnea within 5–7 days. If patients experience a recurrence of apnea, then treatment with caffeine citrate is resumed either at a maintenance dose or at half the loading dose, depending on the time interval that elapses from the moment of discontinuation of caffeine citrate until the moment of recurrence of apnea. Because caffeine is eliminated slowly in this group of patients, there is no need for a gradual dose reduction at the end of treatment. Because there is a risk of recurrence of apnea after discontinuation of caffeine citrate treatment, the patient should be monitored for approximately another week.
Liver and kidney failure
There is limited experience with use in patients with hepatic/renal impairment. In a post-marketing safety study, the incidence of adverse events was higher in a small number of very preterm neonates with hepatic/renal impairment compared with preterm neonates without organ involvement. If renal failure is present, the risk of accumulation increases. The daily maintenance dose of caffeine citrate should be reduced and should be administered only after plasma caffeine concentrations have been determined.
In extremely premature infants, caffeine clearance is independent of liver function. Metabolism of caffeine in the liver develops gradually over several weeks after birth. In older infants with liver disease, determination of plasma caffeine concentrations and dose adjustment may be necessary.
Side effect
Possible adverse reactions of caffeine citrate include:
- stimulation of the central nervous system, manifested by convulsions, irritability, agitation, syndrome of increased neuro-reflex excitability;
- effects on the cardiovascular system with symptoms of tachycardia, arrhythmia, arterial hypertension and increased stroke volume;
- nutritional and metabolic disorders.
These side effects are dose-dependent, so there is a need to determine the concentration of caffeine in the blood plasma and reduce the dose.
Adverse reactions of caffeine citrate published in the literature and obtained in a post-marketing safety study were classified according to WHO recommendations: very common (≥1/10), common (≥1/100 and <1/10), uncommon (≥1/1000 and <1/100), rare (≥1/10000 and <1/1000), very rare (<1/10000), frequency unknown (it is not possible to determine the frequency of occurrence from the available data).
- Infectious and parasitic diseases: frequency unknown - sepsis.
- Immune system disorders: rarely - hypersensitivity reactions.
- Metabolic and nutritional disorders: often - hyperglycemia, frequency unknown - hypoglycemia, poor weight gain, food intolerance.
- Nervous system disorders: infrequently – convulsions, frequency unknown – irritability, syndrome of increased neuro-reflex excitability, excited state, brain damage.
- Hearing and labyrinthine disorders: frequency unknown - deafness.
- Cardiac disorders: often - tachycardia, infrequently - arrhythmia, frequency unknown - increased left ventricular output, increased stroke volume.
- Digestive system disorders: frequency unknown - belching, increased volume of gastric aspirate, necrotizing enterocolitis
- General disorders and disorders at the injection site: often - irritation and inflammation at the infusion site, phlebitis
- Laboratory and instrumental data: frequency unknown - increased sodium and calcium levels in the urine, decreased hemoglobin levels, decreased thyroxine levels.
Description of selected adverse reactions
Necrotizing enterocolitis is a common cause of morbidity and mortality in preterm infants. There is evidence of a possible connection between the use of methylxanthines and the development of necrotizing enterocolitis. However, a cause-and-effect relationship between the administration of caffeine and other methylxanthines and the development of necrotizing enterocolitis has not been established. In a double-blind, placebo-controlled study of caffeine citrate in 85 preterm infants, necrotizing enterocolitis was diagnosed in the blinded phase of the study in 2 infants in the active treatment group and 1 infant in the placebo group; and in the open-label treatment phase in an additional 3 children in the caffeine group. Three children who developed necrotizing enterocolitis during the study died. In a large multicenter study of long-term outcomes in 2006 preterm infants treated with caffeine citrate, there was no increase in the incidence of necrotizing enterocolitis compared with the placebo group. Premature neonates receiving caffeine citrate should be closely monitored for the development of necrotizing enterocolitis. Brain damage, seizures and deafness were also observed, but more frequently in the placebo group. Caffeine may inhibit erythropoietin synthesis, which reduces hemoglobin concentrations during long-term treatment. In infants, at the beginning of therapy, transient cases of decreased thyroxine (T4) were observed, but they disappeared with further therapy. Available data on caffeine treatment do not indicate any damage in neonates with respect to neurological outcome, poor weight gain, cardiovascular, gastrointestinal, or endocrine dysfunction. It is assumed that caffeine cannot aggravate brain hypoxia or enhance existing brain damage, although this possibility cannot be excluded.
Other special populations
In a post-marketing safety study, 506 preterm infants received caffeine citrate. Safety data were obtained in 31 premature newborns with renal/liver failure. The incidence of adverse reactions in a small number of very preterm neonates with renal/liver failure was higher compared with preterm neonates without organ damage. Cardiac disturbances (tachycardia, including one case of arrhythmia) were mainly reported.
Overdose
According to published data, caffeine overdose in premature infants has been observed with plasma concentrations ranging from 50 mg/L to 350 mg/L.
Symptoms
According to the literature, signs and symptoms of caffeine overdose that occur in preterm infants include hyperglycemia, hypokalemia, fine trembling of the extremities, restlessness, hypertension, opisthotonus, tonic-clonic seizures, convulsions, tachypnea (rapid breathing), tachycardia, vomiting, gastric irritation, gastrointestinal bleeding, fever, tremors, increased urea levels in the blood, leukocytosis, involuntary movements of the jaws and lips. One case of caffeine overdose complicated by intraventricular hemorrhage with long-term neurological complications has been reported.
Cases of death of premature newborns associated with caffeine overdose have not been described.
Treatment
Treatment of caffeine overdose is initially symptomatic and supportive. Potassium and glucose levels should be monitored and adjusted if hypokalemia and hyperglycemia occur. Plasma caffeine concentrations have been shown to decrease during exchange transfusion. Seizures can be controlled by intravenous administration of anticonvulsants (diazepam or barbiturates such as sodium phenobarbital or phenobarbital).
Interaction with other drugs
Interconversion of caffeine and theophylline is observed in premature newborns. Therefore, these substances should not be prescribed simultaneously.
Cytochrome P450 (CYP1A2) is the primary enzyme involved in the metabolism of caffeine in adults, and caffeine has the potential to interact with substances that are CYP1A2 substrates, CYP1A2 inhibitors, or CYP1A2 inducers. However, the metabolism of caffeine in preterm infants is limited due to the immaturity of the liver enzyme systems.
Although there is little data on the interaction of caffeine with other active substances in preterm neonates, lower doses of caffeine citrate are given when concomitantly prescribed compounds that reduce the metabolism of caffeine in adults (eg, ketoconazole), and higher doses when co-administered compounds that, on the contrary, enhance its metabolism (for example, phenobarbital and phenytoin). If there is any doubt about the potential interaction of prescribed substances with caffeine, determine the concentration of caffeine in plasma.
Because intestinal bacterial overgrowth is associated with the development of necrotizing enterocolitis, concomitant use of caffeine citrate with drugs that suppress gastric secretions (H2 blockers or proton pump inhibitors) may theoretically increase the likelihood of developing necrotizing enterocolitis.
Coadministration of caffeine and doxapram (a respiratory analeptic) may potentiate their stimulating effects on the cardiorespiratory and central nervous systems. If concomitant use of these drugs is indicated, carefully monitor heart rate and blood pressure.
special instructions
Apnea (stopping breathing)
The diagnosis of primary apnea in premature newborns is made by exclusion. Other causes of apnea (eg, damage to the central nervous system, congenital lung lesions, anemia, sepsis, metabolic disorders, cardiovascular abnormalities, or obstructive apnea) must be excluded/addressed before starting caffeine citrate. Failure to respond to caffeine citrate treatment (confirmed by plasma concentrations, if necessary) may provide evidence that the apnea is due to another cause.
Convulsions
Since caffeine is a central nervous system stimulant, cases of seizures have been reported with caffeine overdose. Particular caution should be exercised when caffeine citrate is administered to newborns with a baseline history of seizure disorders.
Cardiovascular reactions
Caffeine increases heart rate, left ventricular output, and stroke volume. Caffeine citrate should be used with extreme caution in newborns with cardiovascular pathologies. There is a possibility of cases of tachyarrhythmia occurring in predisposed patients. In newborns, this usually manifests as simple sinus tachycardia. If an atypical rhythm disturbance was observed during cardiotocography in the fetus, then caffeine citrate is prescribed with extreme caution.
Kidney and liver failure
The incidence of adverse reactions in very preterm neonates with renal/liver failure was higher compared with preterm neonates without organ damage. In this group of patients, to avoid toxic effects, caffeine doses should be adjusted based on the results of determining plasma caffeine concentrations.
Caffeine consumption
In neonates whose mothers consumed large amounts of caffeine before birth, it is necessary to determine the initial plasma caffeine concentration before starting treatment with caffeine citrate, due to the fact that caffeine readily crosses the placenta into the fetal circulation. Nursing mothers whose infants are being treated with caffeine citrate should not consume caffeine-containing foods, drinks, or medications because caffeine is excreted in breast milk.
Theophylline
In neonates pretreated with theophylline, baseline plasma caffeine concentrations should be determined before initiating caffeine citrate treatment because theophylline is metabolized to caffeine in preterm neonates.
Caffeine citrate may cause increased metabolism, which may result in increased energy and nutrient requirements during treatment.
Diuresis and electrolyte losses caused by caffeine citrate may require correction of fluid and electrolyte imbalances.
In premature newborns, t1/2 of caffeine is prolonged, there is a possibility of drug accumulation, which may require monitoring of newborns receiving this drug for a long time. Blood samples to determine concentrations should be taken immediately before the next dose in cases of lack of therapeutic response to treatment, and only 2-4 hours after the previous dose in cases of suspected toxicity. The range of therapeutic plasma concentrations of caffeine is not indicated, however, in studies showing the clinical effect of caffeine, the concentration ranged from 8 to 30 mg/L; When plasma caffeine concentrations were less than 50 mg/L, no safety concerns were generally encountered.
Sodium
content One dose of Caffeine citrate (1 ml) contains less than 1 mmol of sodium (8.3 mg), which allows the drug to be classified as essentially sodium-free.
Impact on the ability to drive vehicles and operate machinery
Not applicable.
Storage conditions
At a temperature not higher than 25 °C.
Keep out of the reach of children.
Best before date
3 years. Do not use after the expiration date indicated on the package.